Data Entry
PioneerRx
DECEMBER 14, 2022
Take data entry to the next level with PioneerRx’s pharmacy system. PioneerRx allows pharmacies to scan prescription hardcopies to store digital copies in the…
This site uses cookies to improve your experience. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country, we will assume you are from the United States. Select your Cookie Settings or view our Privacy Policy and Terms of Use.
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
Used for the proper function of the website
Used for monitoring website traffic and interactions
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
 Data Entry Related Topics
Data Entry Related Topics 
PioneerRx
DECEMBER 14, 2022
Take data entry to the next level with PioneerRx’s pharmacy system. PioneerRx allows pharmacies to scan prescription hardcopies to store digital copies in the…
Pharmacy Times
APRIL 24, 2024
Carefully checking every piece of information and proofreading for accuracy is crucial to ensure patient safety
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
pharmaphorum
JANUARY 28, 2021
Individualised digital data capture and analytics were enabled by Curelator’s digital technology that combines a simple smartphone data entry process with personalised analytics. After 90 days of data entry, the app creates a trigger, protector and “no association” map for each use within a personalised report.

PharmaTech
OCTOBER 4, 2022
Through the adoption of a data driven and automated CMC Dossier generation process, pharmaceutical companies can reduce time-to-market while delivering significant cost savings to the organization.

National Association of Boards of Pharmacy
FEBRUARY 5, 2024
The Data Quality in Prescription Monitoring Programs course will provide insight into how pharmacy staff data entry processes affect PMP data, clinical decision making, and downstream data analysis.

Pharmafile
APRIL 19, 2023
According to the 2023 Veeva MedTech Clinical Benchmark Report , on-time data entry and data quality are the top challenges for medtech when working with clinical research sites. 2023 — Veeva Systems (NYSE: VEEV) today released its first-ever report examining global trends in medtech clinical trials. read more

Express Pharma
JUNE 11, 2025
Data cleaning, the critical first step? Before scientists can harmonise data, they must clean it. By elim- inating the errors that arise from data entry, miscalculations, sensor malfunctions, or system glitches, scientists can build the data foundation required for harmonisation.

pharmaphorum
JULY 21, 2021
The winning team relied on “handcrafted” features that incorporated their own insights into drug development timelines and which data entries should be discarded, according to MIT.

Flip the Pharmacy
JULY 22, 2024
Their pharmacists count and verify drugs while the technicians focus on data entry including eCare plans. They have a counseling room that is welcoming and stress free for patients to go in and talk with a certified counselor. Bay Street had an abundance of foot traffic in the short time I was there.

Pharma Marketing Network
JUNE 4, 2025
Tasks that are repetitive, data-heavy, or rules-based are most vulnerable. For example, medical data entry, pharmacovigilance documentation, and basic regulatory filing are increasingly handled by intelligent systems. Roles Most at Risk (and Why) Not all pharma jobs face equal threat from automation.

pharmaphorum
SEPTEMBER 16, 2020
Delayed clinical trial data delivery is a growing problem for pharmaceutical companies and one that can have catastrophic consequences for the development of their promising pipeline candidates.

pharmaphorum
SEPTEMBER 24, 2020
Refining data cleansing strategies – Using those data dashboards, Trusts should augment data cleansing and validation strategies to ensure any issues identified in data entry are quickly tackled. It may seem like yet more work to do, and it is.

Express Pharma
JUNE 28, 2023
Features such as updating store inventory details by uploading purchase bills significantly reduce the time in data entry, allowing pharmacists to focus on more complex work. These systems have helped pharmacists efficiently manage their operations, enabling them to generate more revenue.
pharmaphorum
AUGUST 10, 2020
Well-designed forms must: Gather data that’s complete, accurate, and of high quality. Be unambiguous and allow for accurate data entry. Avoid gathering more data than what is needed. Provide form completion guidelines to reduce data capture and data entry issues. Avoid duplication. Get user feedback.
pharmaphorum
AUGUST 3, 2020
Well-designed forms must: Gather data that’s complete, accurate, and of high quality. Be unambiguous and allow for accurate data entry. Avoid gathering more data than what is needed. Provide form completion guidelines to reduce data capture and data entry issues. Avoid duplication. Get user feedback.

Pharmaceutical Technology
APRIL 20, 2023
Combining Rave Imaging with EDC enables the collection of critical data to help manage clinical trials for a unified data platform that saves time, resources and costs. Rave RTSM, which has been developed on Rave EDC, eliminates double data entry and requires reduced reconciliation that speeds up study start-up and close-out.

pharmaphorum
MARCH 5, 2021
The technology known as N1-Headache allows people to use a smartphone to enter data about their symptoms, which it uses to create personalised analytics. After 90 days of data entry, the app creates a trigger, protector and “no association” map for each use within a personalised report.

Pharmaceutical Technology
MAY 31, 2023
It will provide the drug discovery initiative access to advanced algorithms and 230 million big data entries. The Chemiverse platform is useful in all aspects of new drug development, from target discovery to lead compound generation.

Pharma Pathway
FEBRUARY 11, 2023
Note – Candidates with injectables -RnD or complex analytical characterization experience and willing to transition to RA are also welcome (NOT for routine analytical or validation folks) Kindly send your CV to rahul.roy1@viatris.com with the following nomenclature else your profile will not be shortlisted 1.

Pharmaceutical Technology
MARCH 22, 2023
In August of 2022, Fresenius Kabi announced a three-year plan to add GS1 Data Matrix barcodes, also known as 2D barcodes, to its US pharmaceutical portfolio. The aim was to streamline workflows in healthcare facilities by reducing error-prone manual data entry for every customer.

pharmacy mentor
JANUARY 15, 2024
Old Pharmacy vs. New Pharmacy Old vs. New Pharmacy: What Are The Differences Operations Overhaul: The Digital Revolution Old Pharmacy: Manual data entry, reliance on paper folders, sticky notes cluttering desktops, and limited computer use for Patient Medication Records (PMR).

Viseven
DECEMBER 13, 2024
RPA employs software robots, or bots, to carry out tasks such as data entry and extraction, insurance claims processing, insurance verification, payroll calculations, document verification, employee onboarding, and many others. With healthcare RPA, it’s possible to mitigate errors and inaccuracies.
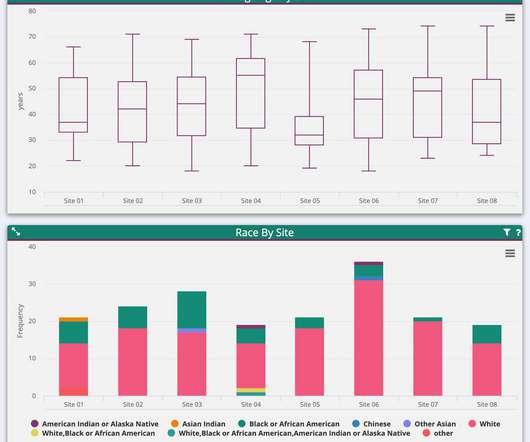
pharmaphorum
SEPTEMBER 11, 2020
Indeed, handling such data and finding resources to clean and manage it was overwhelmingly cited by Pharma Intelligence respondents as the most urgent challenge facing researchers over the next five years. Clearing redundant data then becomes difficult, and programming complex edit checks becomes impossible.

pharmaphorum
DECEMBER 13, 2022
Increased study adherence: Patients are most familiar with their own devices, and the ability to download an application and complete entries on a device they understand increases their adherence to the study requirements. However, in a BYOD study, participants have total control over their devices and can disable reminders themselves.

European Pharmaceutical Review
MAY 22, 2025
2 Reassigning skilled resources : Routine tasks like data-entry and initial case processing are essential but monotonous. AI applications in pharmaceutical technology, from drug formulation to delivery, yield tangible benefits in process optimisation, predictive modeling and decision-making accuracy.

pharmaphorum
OCTOBER 6, 2020
To improve data confidence regulatory executives should take a four-step approach to change how they: Collect Data: Life sciences organisations should utilise cloud-based solutions with global access that facilitates one repository with a single source of truth and eliminates the use of local file sharing and servers.

pharmaphorum
OCTOBER 20, 2022
But by simplifying the basic elements of strategic decision-making and offering a structured process for data entry and analysis, we can ensure teams draw the right insights from the data to inform a strong commercial strategy and bring innovative therapies to market.

European Pharmaceutical Review
SEPTEMBER 26, 2023
For E2B and E2B hybrid migrations, it is important to consider the non-transferable data requirements for the database” If additional non-transferable data is required, a technical migration can be performed, which involves uploading ICSRs into the safety database as electronic E2B files with manual data entry for the additional data.

European Pharmaceutical Review
JULY 29, 2024
Figure 1: A multifaceted approach is needed to achieve data integrity in Biopharma Unlocking the power of automation Automation is often heralded as a panacea for improving efficiency and reducing errors.

pharmacy mentor
SEPTEMBER 16, 2024
Operational Benefits of Wearable Tech in Pharmacies Streamlining Workflow Automated Data Collection : Wearable devices automatically collect health data, reducing the need for manual data entry and allowing pharmacists to focus on patient care.

pharmacy mentor
SEPTEMBER 16, 2024
Operational Benefits of Wearable Tech in Pharmacies Streamlining Workflow Automated Data Collection : Wearable devices automatically collect health data, reducing the need for manual data entry and allowing pharmacists to focus on patient care.

National Association of Boards of Pharmacy
APRIL 10, 2024
Then, walk through each step of the practice of pharmacy such as data entry; drug utilization review; compounding; final association between the drug, the prescription, and the label (eg, the dispensing act); and patient counseling.

pharmaphorum
DECEMBER 6, 2022
In most cases, the EHRs and EDCs don’t communicate, so in order to share that data with trial organisers, staff members at medical centres must manually copy data between the EHR and an EDC. As an average Phase 3 trial amasses over 3.6

PharmaState Academy
MARCH 4, 2024
Option 02 Increasing paperwork and manual data entry. Read the whole case study scenario Close Brainstorming Question What is a key benefit of leveraging technology in pharmaceutical sales? Option 01 Automating routine tasks and freeing up time for strategic sales activities.

European Pharmaceutical Review
MARCH 12, 2024
Implementing effective measures to mitigate the risk of human error includes establishing error-checking procedures, providing clear and concise instructions, and establishing systems for double-checking critical data entries. Human errors, whether intentional or unintentional, can have significant implications for data integrity.

FADIC
JULY 10, 2023
With Quinvio AI, businesses can eliminate the manual and time-consuming process of data entry and processing invoices, enabling them to focus on more strategic and valuable tasks. can analyse and optimise data, ensuring that it is accurate, up-to-date, and easy to interpret.
RX Note
NOVEMBER 13, 2024
Hence, we hope all report things can be auto-calculated with each new data entry. Ctrl + Shift to Select To quickly select a dataset, you can click in the first cell that you want to use and hold down Ctrl+Shift , then hit Down arrow or Up arrow to get all the data below or above in the column.

PharmaShots
JUNE 6, 2023
In such cases, companies may initially begin by infusing small aspects of Robotic Process Automation (RPA) into essential areas like R&D, clinical trials, and patient data entry. For more information email us at connect@pharmashots.com References: Javatpoint EMERJ P360

Viseven
SEPTEMBER 6, 2024
Data integrity isn’t a software, service, or product; it encompasses various solutions contributing to improved data maintenance and quality. Importance of Data Integrity in the Pharma Industry It takes just one wrong data entry, breach, or incident for patients and clients to lose your trust.

Pharmaceutical Technology
JUNE 15, 2023
5 examples of AI for clinical trials By using AI-driven algorithms for a wide range of tasks, from data entry and analysis to improved medical diagnoses, companies can reduce the time and cost associated with drug development. Here are some ways companies are using AI in clinical trials.

GMPSOP
OCTOBER 16, 2022
A process to prohibit handwritten GMP documents except for handwritten approval entries for master documents or handwritten data entries on records ; iv. A process that prohibits conflicting or redundant information among documents; iii. Defined responsibilities for review and approval of documents at the site; v.

GMPSOP
APRIL 19, 2024
Excel spreadsheets are widely used in pharmaceutical operations, from simple data entry in the registers to complex data processing using functions and formulas. This would involve typical day-to-day tasks, such as data entry, calculations, generating reports, or any other regular function the spreadsheet is intended for.

GMPSOP
OCTOBER 28, 2023
. – Record or capture all generated raw data directly, promptly, and legibly. – Use traceable data sheets or sequentially numbered notebooks. – Date and sign or initial data entries on the day of entry. – Archive records so that they are protected, secured, and easily retrievable.

GMPSOP
DECEMBER 24, 2023
. – Record or capture all generated raw data directly, promptly and legibly. – Use traceable data sheets or sequentially numbered notebooks. – Date and sign or initial data entries on the day of entry. – Archive records so that they are protected, secure, and easily retrievable.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content