Diabetes Awareness Month 2019
PioneerRx
DECEMBER 14, 2022
November is National Awareness Diabetes Month - keep reading to see how community pharmacists play an important role in the prevention of diabetes and what you can…
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

PioneerRx
DECEMBER 14, 2022
November is National Awareness Diabetes Month - keep reading to see how community pharmacists play an important role in the prevention of diabetes and what you can…

Pharmaceutical Technology
MAY 3, 2024
Despite failing to get an approval for type 1 diabetes in 2019, Lexicon plans to resubmit its NDA to the FDA by mid-2024.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
STAT
NOVEMBER 18, 2022
The first therapy that delays the onset of type 1 diabetes received approval from the U.S. As of 2019, about 1.9 million people have type 1 diabetes in the U.S., according to the American Diabetes Association, including 244,000 children and adolescents. Type 1 affects 8% of everyone with diabetes.

European Pharmaceutical Review
MARCH 8, 2024
Approximately 40 percent of people with type 2 diabetes have chronic kidney disease, so the positive results from FLOW demonstrate the potential for semaglutide to become the first GLP-1 treatment option for people living with type 2 diabetes and chronic kidney disease.”

PharmExec
MARCH 14, 2024
Resubmission plan comes amid feedback and a complete response letter issued by the FDA in 2019 for sotagliflozin in the treatment of glycemic control in type 1 diabetes patients with chronic kidney disease.

Express Pharma
APRIL 27, 2023
Cipla holds a prominent position in the top five therapy areas in India, except for the anti-diabetes and gastrointestinal segment. Against this backdrop, the Indian pharma major is constantly partnering with multinational companies to get access to an innovative portfolio with a focus on diabetes, says GlobalData.

STAT
FEBRUARY 8, 2023
People with diabetes, high blood pressure, and other metabolic conditions are at especially high risk of developing the condition. It’s an especially costly condition to treat — in 2019, Medicare spent more than $87.2

pharmaphorum
MARCH 11, 2021
Vertex Pharma has made its name with treatment for cystic fibrosis, but has big plans in cell and genetic therapies – and has just chalked up fast-track status from the FDA for a cell-based therapy for diabetes. . The aim is to replace the pancreatic tissue that is damaged by the autoimmune reaction that underlies type 1 diabetes.

pharmaphorum
DECEMBER 21, 2022
Sosei Heptares’ longstanding alliance with Pfizer has advanced a first candidate into phase 2 testing: an oral GLP-1 agonist for diabetes and obesity that will try to take on established rivals from Novo Nordisk and Eli Lilly. The post Sosei nets $10m Pfizer milestone as diabetes/obesity drug starts phase 2 appeared first on.

Viseven
MARCH 27, 2023
In February 2019, Viseven, a global provider of digital solutions for the life sciences industry, announced a strategic collaboration with a global pharmaceutical company. This company specializes in biopharmaceuticals, diabetes care, obesity care, and other severe chronic diseases. Let us know!
STAT
SEPTEMBER 23, 2022
These 22 products compete in nine molecule classes across oncology, rheumatology, diabetes care, and now ophthalmology. billion in 2020 (three times higher than savings from 2019) and have the potential to increase to $133 billion by 2025. According to the most recent “U.S.

pharmaphorum
SEPTEMBER 17, 2020
Taiwan-based digital health firm Health2Sync has partnered with Sanofi to help patients manage symptoms of diabetes. While Sanofi is winding up its R&D operations in diabetes it is turning to digital solutions to get the most from its established products for the disease. million diabetes patients in Taiwan.

pharmaphorum
JULY 11, 2022
Vertex has doubled down on cell-based therapies for type 1 diabetes (T1D), buying ViaCyte in a $320 million deal that it says will accelerate development of its own candidate VX-880. The post Vertex buys diabetes cell therapy rival ViaCyte for $320m appeared first on.

pharmaphorum
NOVEMBER 30, 2021
Francine Kaufman discusses how Senseonics is helping patients monitor their diabetes with the first fully implantable long-term continuous glucose monitoring (CGM) system. Kaufman has been a practicing pediatric endocrinologist with a focus on diabetes for almost 45 years. She joined the team at Senseonics in 2019.
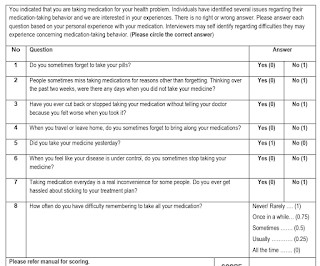
RX Note
APRIL 10, 2024
Malaysia Medication Adherence Assessment Tool (MyMAAT) The Malaysia Medication Adherence Assessment Tool (MyMAAT) was developed by a multidisciplinary team with expertise in medication adherence and health literacy for use with diabetic patients. External Links Pay up or retract?

The Guardian - Pharmaceutical Industry
JANUARY 10, 2023
Researchers find overall percentage increase from 0.06% in 2000 to 0.11% in 2019 although safety in children not fully established The proportion of children and young people prescribed antipsychotics in England has nearly doubled in just two decades, prompting concern among some experts. Continue reading.

pharmaphorum
APRIL 13, 2022
It has now been 100 years since the first patient with diabetes received an injection of insulin, when Leonard Thomas, a 14-year-old boy, was treated with the hormone. In recent years, there have been more developments in the area of diabetes management, with the emergence of metformin and more recently semaglutide and dulaglutide.
Express Pharma
FEBRUARY 18, 2024
In a recent quarterly update, AstraZeneca revealed that it removed tozorakimab from its Phase II pipeline for diabetic kidney disease (DKD). AstraZeneca progressed tozorakimab into Phase IIb for DKD in 2019, as there was evidence that increased inflammation is associated with deterioration in renal function.

STAT
MARCH 26, 2024
a decade after the chain announced it would implement a “No Antibiotics Ever” policy by 2019. Philadelphia District Attorney Larry Krasner is suing insulin makers and pharmacy benefit managers that he alleges have artificially inflated the cost of a life-saving diabetes medication , Philly Voice tells us.
Pharmaceutical Technology
APRIL 25, 2023
Baqsimi is the first and only nasally given glucagon to treat very low blood sugar, or severe hypoglycaemia, in diabetes patients. Lilly launched the drug in 2019 to provide rescue treatment for diabetes patients who take insulin. It is now available in 27 international markets, with $139.3m in global sales reported last year.

Drug Channels
JULY 29, 2020
In this issue: Pricing problems for diabetes treatments Controversy over Regeneron’s copay support Massachusetts concedes that coupons help some patients (but accumulators hurt) A 2020 update to my all-time favorite chart Plus, thoughtful perspectives on diversity from AmerisourceBergen CEO Steve Collis.
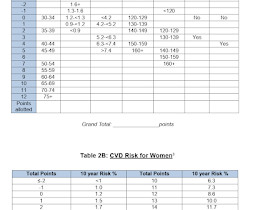
RX Note
JUNE 23, 2023
The common cardiovascular risk factors include: Dyslipidemia Hypertension Diabetes Mellitus Smoking Overweight/obesity Dyslipidemia Dyslipidemia refers to the following lipid levels: Total cholesterol (TC) > 5.2 Patients with established CVD, CKD and diabetes fall into the very high and high risk categories.
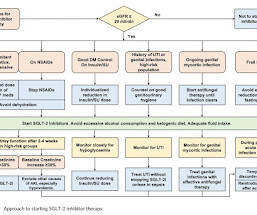
RX Note
JUNE 10, 2023
Introduction The treatment approach for type 2 diabetes mellitus has undergo a shift from the traditional glucose-centric approach to the new cardiorenal-metabolic approach. This change emphasizes a comprehensive approach for reducing the risk of diabetes-related complications.

Pharma Mirror
JANUARY 24, 2023
With the promise of significant breakthroughs in treatment of diabetes, cancer, and congenital conditions, it is no wonder that research and development is predicted to reach $232.5 billion by 2026, up from $186 billion in 2019 according to the Evaluate Pharma report.
Med Ed 101
DECEMBER 17, 2023
Data from poison control centers across the country indicates a drastic increase over the past year in calls related to semaglutide overdose — an increase of over 1500% since 2019 according to a […] The post Semaglutide Overdose – What Does It Look Like and Why Is It Happening? appeared first on Med Ed 101.
pharmaphorum
JANUARY 24, 2022
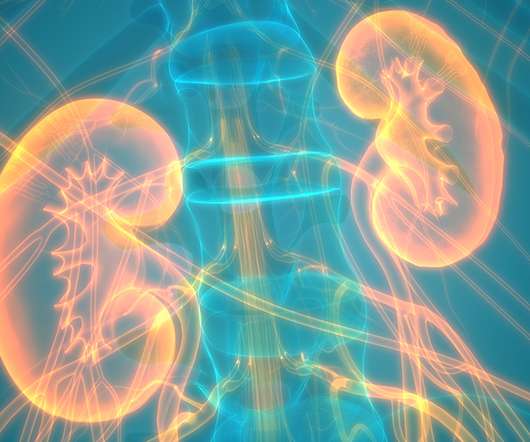
In November, NICE backed NHS use of AstraZeneca’s diabetes and heart failure drug Forxiga for the treatment of chronic kidney disease. It was initially developed for type 2 diabetes and was approved for this indication in 2014. The DAPA-HF and DAPA-CKD trials were launched, including patients with and without type 2 diabetes.

RX Note
MARCH 8, 2024
External Links NPRA - Metformin: Risk of Vitamin B12 Deficiency, 2022 [Updated] Metformin: Risk of Vitamin B12 Deficiency, 2024 MHRA - Metformin and reduced vitamin B12 levels: new advice for monitoring patients at risk Association between metformin dose and vitamin B12 deficiency in patients with type 2 diabetes, 2019 Prevalence of Vitamin B12 Deficiency (..)

pharmaphorum
JANUARY 6, 2021
Danish drugmaker Novo Nordisk signed a deal with the US biotech in 2019 valued at up to $675.5 million – including $175 million upfront and an equity investment of $50 million – to find multiple RNA interference (RNAi) candidates for diseases like non-alcoholic steatohepatitis (NASH) and type 2 diabetes.
pharmaphorum
JANUARY 12, 2021
Boehringer and Eli Lilly have moved closer to a heart failure indication for their SGLT2 inhibitor Jardiance, as the FDA starts a fast-track review of the drug in its first use beyond diabetes. Invokana was the first mover among the SGLT2 drugs in the kidney area, winning FDA approval towards the end of 2019 for diabetic kidney disease.

pharmaphorum
NOVEMBER 16, 2022
Novartis completed the spin-out of Alcon in 2019, separating products like contact lenses and over-the-counter eyedrops from its prescription eye medicines, headed by age-related macular degeneration (AMD) blockbuster Lucentis (ranibizumab), which is partnered with Roche. billion in the first nine months of this year.
RX Note
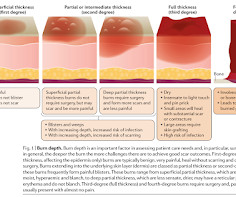
DECEMBER 26, 2023
In diabetes, a burn on a foot, even mild, could lead to an amputation. External Links Burn injury, 2020 Mayo Clinic - Burns The Effect of Aloe Vera Clinical Trials on Prevention and Healing of Skin Wound: A Systematic Review, 2019 Vigilance is required. Initial Management Most minor burns can be treated at home.
pharmaphorum
JULY 29, 2021
The US regulator will review the drug for both neovascular or ‘wet’ age-related macular degeneration (AMD) and diabetic macular oedema (DME), as well as diabetic retinopathy under a standard review. Beovu launched in October 2019 in the US with sales predictions of $2.5

Big Molecule Watch
DECEMBER 14, 2023
AVZIZI is the fifth bevacizumab biosimilar approved by the FDA, behind Amgen’s MVASI (bevacizumab-awwb), approved in September 2017; Pfizer’s ZIRABEV (bevacizumab-bvzr), approved in June 2019; Amneal’s ALYMSYS (bevacizumab-maly), approved in April 2022; and Celltrion’s VEGZELMA (bevacizumab-adcd), approved in September 2022.

pharmaphorum
JANUARY 13, 2023
.” He added that people from low-income households and some racial and ethnic groups are disproportionately impacted by the practices, including Hispanic and Black people, who are much more likely to be diagnosed with diabetes than non-Hispanic white people – and much more likely to die as a result of complications.

Express Pharma
DECEMBER 18, 2023
Shweta joined Bayer in 2019 and her last assignment was Business Unit Head. Her expertise extends across a myriad of therapy areas, including Cardiology, Diabetes, Women’s Health Care, Immunology, Virology, Anti-infectives, Vaccines, Neurology, Orthopedics and Pain Management.

PharmD Live
DECEMBER 21, 2021
In 2019 and 2020, there is a measurable difference in how televisits increased in underserved populations, specifically Medicaid, Medicare and minorities. Insulin, first introduced in 1921, 100 years later, 2021, diabetes monitoring devices are added to Medicare’s durable medical equipment list coverage. Aduhelm earns FDA approval.

The Thyroid Pharmacist
SEPTEMBER 2, 2022
A review paper published in 2010 that discussed the relationship between thyroid dysfunction and metabolic disorders, highlighted the fact that patients with diabetes were up to three times more likely to develop a thyroid disorder. (7). Hypoglycemia is an early risk factor for developing diabetes. Type 2 Diabetes.

pharmaphorum
DECEMBER 1, 2020
First results of the DiabetRisk study – Over 8% of patients identified with undiagnosed diabetes or pre-diabetes at dental practices. The DiabetRisk study could contribute to early diagnosis of diabetes in thousands of dental patients each year – and in every country worldwide.
pharmaphorum
JUNE 24, 2021
Roche bought ex-US rights to the PDS version from Novartis in 2019, giving it total control of the implant product, while Novartis has focused on the development of longer-acting VEGF antibody Beovu (brolucizumab), which is dosed every two or three months. It launched in October 2019 in the US with sales predictions of $2.5

The Guardian - Pharmaceutical Industry
JULY 8, 2023
Drug firms are simultaneously funding groups that lobby for greater investment in their disease areas, and in some cases are paying generous consultancy fees to influential healthcare professionals, including GPs who have worked as clinical leads for NHS England and have received as much as £480,000 each from industry since 2019.

RX Note
MAY 31, 2023
Introduction In 2017, the Malaysian Diabetes Educators Society published the Forum for Injection Technique Malaysia (FIT-MY) to provide evidence-based injection technique recommendations for people with diabetes mellitus who use injectable therapy in their daily management. If you have not, repeat steps above.
STAT
JULY 10, 2023
were prescribed a GLP-1 agonist in 2022, up from about 230,000 in 2019, according to a recent analysis by data insights company Komodo Health. Around Thanksgiving 2021, Fernandez’s doctor approached him about trying something different: a new weight loss drug called Wegovy.

pharmaphorum
JULY 27, 2021
Most recently, the French company has secured US and European marketing approvals for a new version of Insulia, a DTx designed for use by people with type 2 diabetes to help them titrate to the correct dose of insulin. Oleena was approved in the US in 2019 and in Europe the following year.

PharmaState Academy
MAY 18, 2023
Merck, and Abbott handling the Local as well as global brand specialists for India for Cardiology & diabetes portfolio respectively. She has also presided at various pharma management colleges as a guest lecturer, Judge & Guest of Honour since 2019 Ms. She has worked with some leading companies namely Sun Pharma, E.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content