Top 3 Manufacturing and Packaging Articles of 2023
Pharmaceutical Commerce
DECEMBER 29, 2023
Popular works in the category explore 340B guidelines, ‘smart pharma’ capabilities, and commercialization trends.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharmaceutical Commerce
DECEMBER 29, 2023
Popular works in the category explore 340B guidelines, ‘smart pharma’ capabilities, and commercialization trends.

European Pharmaceutical Review
APRIL 19, 2023
A market report has identified that global demand for electronic smart packaging will reach $2.6 percent from 2023, according to the data. Smart packaging has potential for the integration of electronic functionality, enabling compliance, material identification, condition monitoring and asset tracking of pharmaceutical products.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Safe Biologics
NOVEMBER 12, 2023
6 would prevent the HHS Secretary from requiring a switching study as part of the data package to receive the interchangeable designation. Register for the 2023 National Policy & Advocacy Summit on Biologics here. However, S.6 ASBM surveys of U.S. Read the new FDA’ Guidance here. Submit comments on the guidance here.

Safe Biologics
DECEMBER 6, 2023
REMINDER: FDA Draft Guidance Would Remove Interchangeability Statement from Interchangeable Biosimilars Comments Due November 17th On September 15th, the Food and Drug Administration (FDA) released draft guidance removing the interchangeability statement from the product label/package insert. ASBM surveys of U.S. However, S.6
European Pharmaceutical Review
FEBRUARY 2, 2023
The winners of Pharmapack Europe’s 2023 awards have been announced, presented across a range of innovative recyclable packaging, drug delivery solutions, and reusable connected devices. The winner in the Eco-Design category was a high grade, 100 percent PET blister packaging.
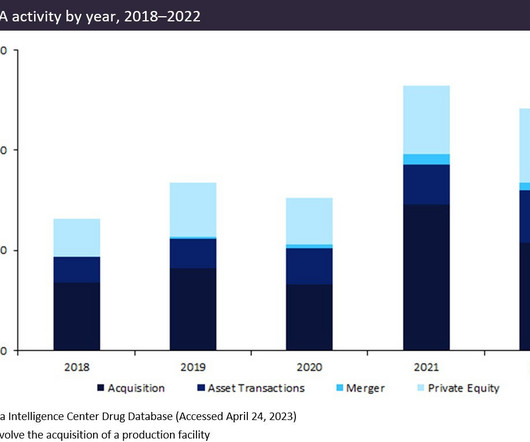
Pharmaceutical Technology
MAY 26, 2023
Pharma company merger and acquisition (M&A) activity in the first half of 2023 has remained relatively low, consistent with the second half of last year, but investors predict deals will increase later in 2023. Experts at PWC, Goldman Sachs, and Morgan Stanley agree that pharma-related M&A is expected to pick up in 2023.

Express Pharma
DECEMBER 11, 2023
So, in this article, we delve into this dynamic market to explore the driving factors behind its exponential growth and the trends shaping the market landscape while evaluating the pain points that need to be addressed for sustained growth. per cent from 2023 to 2033, compared to 13.3 billion in 2022. per cent during 2022-2028.

European Pharmaceutical Review
JANUARY 25, 2024
In a survey conducted by The European Consumer Organisation (BEUC) during June and July 2022, in Belgium, Italy, Portugal and Spain, 79 percent of consumers said paper leaflets should be available inside the package even if there is an alternative QR code on it. 11 This requirement directly contradicts what is included in Article 63.

Pharmacy Joe
OCTOBER 25, 2023
The removal appears to be in response to the publication of a 2021 article in the International Journal of Pharmaceutical Compounding. It appears that Pharmacist’s Letter has stepped in to fill the need for this list as in February 2023 they published and updated a list of Meds That Should Not Be Crushed.

Express Pharma
DECEMBER 7, 2023
” After the resounding success of Ahmedabad Pharma Summit 2023, Express Pharma recently hosted the fourth edition of Vizag Pharma Summit on May 19, 2023 at the Hotel Taj Gateway in Vishakhapatnam. Express Pharma ’s next ‘Pharma Summit’ will be held in Chandigarh on June 30, 2023 at The Holiday Inn, Panchkula.

GMPSOP
DECEMBER 10, 2023
Particularly the printed packaging materials where product information is presented. Printed packaging materials typically include product names, active ingredients, concentration, batch numbers, expiry dates, registration numbers, barcodes, etc. Commonly used packaging materials are cartons, inserts, leaflets, printed foil, etc.

Pharma Marketing Network
MARCH 25, 2023
This includes everything from the company’s website and social media presence to packaging and advertising. ” Entrepreneur, www.entrepreneur.com/article/314384. All communications, both internal and external, should reflect the brand’s values and personality. References: 1. ” Communiqué, communique.us/creating-strong-brand-identity-pharmaceutical-industry/.

Pharmacy Joe
OCTOBER 25, 2023
The removal appears to be in response to the publication of a 2021 article in the International Journal of Pharmaceutical Compounding. It appears that Pharmacist’s Letter has stepped in to fill the need for this list as in February 2023 they published and updated a list of Meds That Should Not Be Crushed.

FDA Law Blog: Biosimilars
JANUARY 3, 2024
Specifically, CVM ensures that animal drugs are safe and effective, properly made, and adequately labeled and packaged; food-producing animals only take drugs that would be safe for humans to consume; pet foods and additives are safe; and educates the public, monitors the market, and encourages development of new animal health products.

epicur
DECEMBER 5, 2022
billion in 2020 (Packaged Facts) Pet medication sales have grown at a healthy CAGR of 9% since 2017 (Packaged Facts) Veterinary practices are unique in that they can sell medications directly to their clients, which provides an important revenue stream for the business. Bn in 2020 to over US$ 2.7 Bn in 2020 to over US$ 2.7

National Association of Boards of Pharmacy
DECEMBER 6, 2023
link] How the Government is Addressing Xylazine Regulation In spring 2023, Congress introduced the Combating Illicit Xylazine Act (S 993/HR 1839). Further, FDA’s Center for Veterinary Medicine and CBP are working to identify xylazine shipments that are packaged and declared other products.
GMPSOP
NOVEMBER 19, 2023
The scope of this article applies to all regulated medicinal products manufactured at a GMP site that undergo annual product quality review. The content of this article does not supersede or replace any local or international regulatory requirements. Subscribe f.

RX Note
MARCH 23, 2023
Today, stability information can be obtained from manufacturer’s labelling information (such as in the package insert), the USP compounding monographs, or peer-reviewed articles and references , such as articles in American Journal of Health-System Pharmacy, the International Journal of Pharmaceutical Compounding.

Viseven
JUNE 12, 2023
In this article, we’ll focus on a pharmaceutical market in the APAC region, go through the differences between pharma in APAC and pharma in other parts of the world, and see the current state of the APAC pharma market. The global pharmaceutical market is becoming more digitalized, insight-driven, and consumer-centric.

Express Pharma
DECEMBER 11, 2023
The latest in this series, Chandigarh Pharma Summit 2023 was successfully hosted by Express Pharma recently. In this article, we delve into the highlights of the Chandigarh Pharma Summit 2023, and showcase the key takeaways and insights from the presentations and discussions at the event.

European Pharmaceutical Review
APRIL 21, 2023
The leaked draft of the European pharmaceutical package is promising on this point as it states that marketing authorisation may be refused if the ERA is not completed or risk mitigation measures are insufficient, and that an ERA programme should be set up for medicines that were authorised before the introduction of the ERA.

European Pharmaceutical Review
FEBRUARY 7, 2023
A wave of important legislative changes is anticipated in the EU in 2023 making this year one of the biggest for the EU regulatory landscape governing the life sciences industry in recent times. As a key priority action under the ACT EU initiative, EU regulators plan to establish a multi-stakeholder platform (MSP) in 2023.

Fuld & Company Blog
MAY 15, 2023
Technology was used to handle costly administration issues. The Resulting Package By 2020, it was clear that One Medical was hitting a positive note with the market, and so it went public. Additionally, in January 2023 Amazon launched RxPass, a $5 monthly subscription plan for U.S. Prime and, of course, One Medical members.
Quality Matters
JUNE 14, 2023
An expanded rationale for the revision was included in a related Stimuli article. Other general chapters are also in development that cover topics such as the qualification of packaging systems for distribution, lane mapping, and information systems for distribution validation/verification studies.

Birth Control Pharmacist
SEPTEMBER 20, 2021
While not stated in the current package insert, data are emerging that support fewer breast tissue effects and breakthrough bleeding than older oral contraceptives. Nextstellis (Drospirenone and Estetrol) [package insert]. OUP Academic , Oxford University Press, 5 July 2014, academic.oup.com/carcin/article/35/11/2447/416699.

Express Pharma
JANUARY 12, 2024
Further, introduction of the Scheme for Promotion of R&D and Innovation in Pharma Med Tech Sector (PRIP) in September 2023 showcases the focus on creating an ecosystem for innovation and research. The concessional rate should not be restricted to only research in relation to article/thing manufactured or produced by the company.
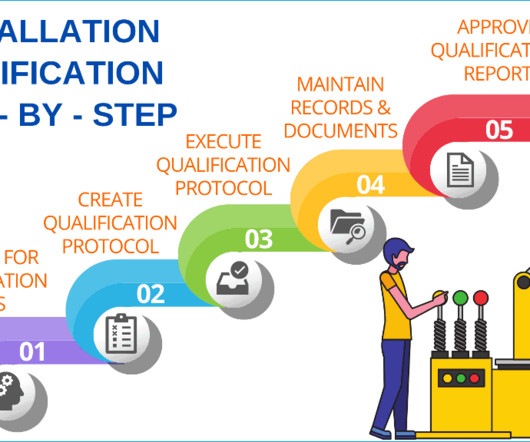
GMPSOP
JULY 23, 2023
If you are completely unfamiliar with what is installation qualification then we suggest you read the article Validation in Pharmaceutical Industry first. If you would like to know more on how to prepare acceptance criteria to use using verification of common pharmaceuticals equipment please read our article on operational qualification.

Fuld & Company Blog
JULY 31, 2023
Tesla Energy Strategy In this article, we investigate Tesla Energy’s overall strategy from making renewable solar energy to storing, selling and trading it, encompassing solar energy production, energy storage, and electricity sales and trading.

GMPSOP
DECEMBER 24, 2023
This article will focus on G(C)LP due to its significance in everyday laboratory operations. This method validation package should confirm the suitability of the method. Understanding GLP, G(C)LP and GCP GLP : Good Laboratory Practices refer to the rules concerning testing new drugs before conducting clinical trials.

Proxsys Rx
JANUARY 12, 2024
Between late 2019 and the end of 2023, ProxsysRx’s 340B Support program went from Zero to $435 million in savings generated for the hospitals and health systems we serve. For a start on ensuring that your 340B program is maintaining best practices, click here to access our in-depth article on the topic. What we’ve learned.

Proxsys Rx
APRIL 19, 2024
Between late 2019 and the end of 2023, ProxsysRx’s 340B Support program went from Zero to more than $500 million in savings generated for the hospitals and health systems we serve. For a start on ensuring that your 340B program is maintaining best practices, click here to access our in-depth article on the topic. What we’ve learned.

Quality Matters
FEBRUARY 13, 2023
DMFs are submissions to the FDA used to provide confidential, detailed information about facilities, processes, or articles used in the manufacturing, processing, packaging, and storing of human drug products. kristine.gates… Mon, 02/13/2023 - 11:24 Supply Chain Read more insights from USP’s Medicine Supply Map.

GMPSOP
OCTOBER 28, 2023
Packaging material specifications In the pharmaceutical industry, the requirements for pre-printed packaging materials must be well-defined and documented to ensure that you get the items you specified or ordered and that there are no mix-ups. A standard name and a unique item code define packaging materials.

ID Stewardship
NOVEMBER 29, 2023
In this article the host of the Let’s Talk Micro Podcast (Luis Plaza Rios) is interviewed about his experience and learning clinical microbiology using podcasting via the Let’s Talk Micro podcast. I had the pleasure to meet Luis in person and after exchanging some emails we were able to compose this article.

GMPSOP
JULY 14, 2023
In this article, we will discuss important aspects of equipment cleaning that are required during pharmaceutical manufacturing. Equipment cleaning validation protocol was not discussed intensely in this article which was covered elsewhere in this blog.

Fuld & Company Blog
APRIL 6, 2023
Today, in 2023, Tesla is reported to have installed 430,000 solar systems across the US. Total sales of energy generation and storage are already estimated to potentially reach $12 billion in 2023. This includes trading and microgrid packages. Tesla and Virtual Power Plants However, the story doesn’t end there.
European Pharmaceutical Review
FEBRUARY 23, 2023
This article presents an overview of Raman spectroscopy and details some of the developments of interest to the pharmaceutical sector. The potential of Raman for microbiology The current advantages and future potential of Raman spectroscopy as a bacterial identification and enumeration method have been outlined in this article.
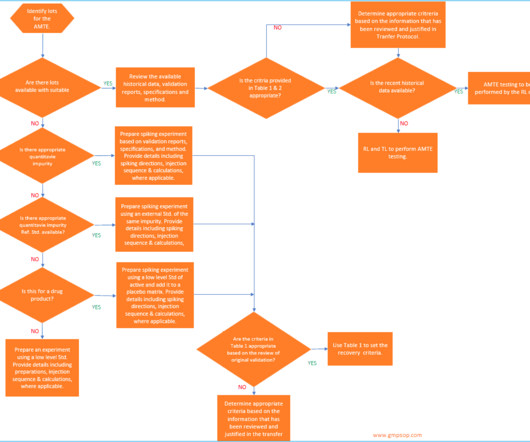
GMPSOP
JUNE 16, 2023
Importance of acceptance criteria in analytical method transfer Pharmaceuticals quality assurance & validation procedures GMPSOP Importance of acceptance criteria in analytical method transfer Last modified: June 17, 2023 Table of Contents What is analytical method transfer?

The Thyroid Pharmacist
NOVEMBER 11, 2022
In this article, I share some fun ideas for gifts you can give (or add to your own wishlist ;-)). Your 2023 health goals. Check out my article on How Adaptogenic Herbs Benefit Hashimoto’s to learn more.). But being health conscious doesn’t mean we can’t give or receive fun holiday gifts! You’ll find gift ideas for: Relaxation.

The Thyroid Pharmacist
OCTOBER 27, 2023
3] You can learn more about the HPA axis and adrenals here in the linked article. Follow package instructions. Follow package directions for each supplement. Doses of 300 mg are generally enough for everyday support, but you may wish to increase your dose for the situations listed above, per package instructions.

The Thyroid Pharmacist
MARCH 10, 2023
I’ve focused a great deal on resolving this with my previous articles on resolving fatigue. I will cover the fundamentals of sleep hygiene for completeness’ sake, but if you’ve been doing ALL of the “basic” things and are still not sleeping, this article will hopefully provide you with additional root cause relief.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content