FDA rejects label expansion for Dynavax’s hepatitis B vaccine
Pharmaceutical Technology
MAY 15, 2024
The US agency rejected the expanded use of Dynavax’s vaccine for adults on haemodialysis, citing insufficient efficacy and safety data.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharmaceutical Technology
MAY 15, 2024
The US agency rejected the expanded use of Dynavax’s vaccine for adults on haemodialysis, citing insufficient efficacy and safety data.
NY Times
APRIL 20, 2021
The European Union’s drug regulator said that the Johnson and Johnson vaccine should carry a warning of potential risk for rare blood clots on the label, but did not recommend stopping the vaccine, saying the benefits of the vaccine outweighed the risks.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Express Pharma
MAY 11, 2023
TechInvention Lifecare India, has partnered with Eubiologics Co, South Korea, to launch Euvichol-Plus, a oral cholera vaccine (OCV), in a low-density polyethylene (LDPE) unidose pack in India. The post TechInvention launches Euvichol-Plus, oral cholera vaccine in India appeared first on Express Pharma.

FDA Law Blog: Biosimilars
AUGUST 21, 2023
Livornese — I saw the sign…and the answer is no—FDA-approved labeling apparently is not enough under state failure-to-warn laws, according to certain courts. A recent state law failure-to-warn case in the SDNY makes that very point.
Pharma Times
JULY 25, 2022
Imvanex to include protection from monkeypox and diseases caused by vaccinia virus

pharmaphorum
MAY 10, 2021
A review of side effects reported with coronavirus vaccines by the EMA’s safety committee has uncovered cases of inflammation of the heart in people receiving the Pfizer/BioNTech Comirnaty shot. . It is also asking for similar data from Moderna, which manufacture a COVID-19 vaccine that like Comirnaty is based on mRNA.

pharmaphorum
FEBRUARY 9, 2022
In this article, Ben Hargreaves looks into the promise of cancer vaccines and how this treatment modality may offer advantages over existing immunotherapies in the oncology sector. One area that is gathering increasing levels of interest is the development of cancer vaccines. A broad front.

pharmaphorum
AUGUST 25, 2021
Just a month after getting approval for its new pneumococcal vaccine Vaxneuvance in adults, Merck & Co has reported positive trial results in children that will ramp up the pressure on Pfizer and its market-leading Prevnar franchise.

pharmaphorum
JANUARY 28, 2021
The Covid-19 pandemic has put further pressure on clinical trial suppliers to be fleet of foot in getting products dispatched when a new patient is recruited, as studies for vaccines have been set up and run at unprecedented speed. . To find out more about the webinar and to register, visit [link] .

Quality Matters
FEBRUARY 9, 2023
Africa’s growing vaccine capacity Manufacturing products locally for more equitable pandemic responses Every year, vaccines prevent nearly 5 million deaths around the globe. Yet, Africa produces less than 1 percent of the vaccines it needs to protect patients and communities across the continent.

Pharmaceutical Technology
JULY 25, 2022
The European Commission (EC) has extended Bavarian Nordic ’s smallpox vaccine Imvanex (MVA-BN or Modified Vaccinia Ankara-Bavarian Nordic) marketing authorisation to offer protection from monkeypox. A non-replicating smallpox vaccine, Imvanex was developed in partnership with the US Government.

pharmaphorum
DECEMBER 16, 2022
Ben Hargreaves discovers why some have referred to the distribution of COVID-19 vaccines and treatments as a form of apartheid. Vaccine access for a price. Tendayi Achium, labelled the response by the global community as a form of “vaccine apartheid,” a suggestion echoed by the organisation’s director general.
pharmaphorum
MAY 26, 2021
Belgium’s Ministry of Health has paused dosing of people under the age of 41 with Johnson & Johnson’s one-shot COVID-19 vaccine, following the death of a woman from what appeared to be a blood clot-related condition. . Last month, the EU regulator concluded that the benefits of the vaccine still outweigh any risks.
Pharmaceutical Technology
OCTOBER 26, 2023
GSK plans to submit the data to regulatory agencies to support label expansion for Arexvy in the younger adult patient population in 2024.
pharmaphorum
MARCH 12, 2021
EU regulators are reviewing reports of low blood platelets in patients who received any of the three approved COVID-19 vaccines from Pfizer/BioNTech, AstraZeneca and Moderna. The PRAC will investigate and if a causal relationship is confirmed or considered likely, an update to the products’ labels will be the most likely outcome.

Pharma Packaging Solutions
NOVEMBER 8, 2023
Pre-filled Syringe Packaging A lot of biopharmaceutical products—such as insulin, vaccines, and many hormone therapies—are parenterally administered. Vial and Ampoule Labeling Many biologics are available primarily in liquid form. Tjoapack handles crucial secondary packaging jobs like vial and ampoule labeling.
pharmaphorum
JULY 9, 2021
The EU medicines regulator has said that there is some evidence to suggest a possible link between mRNA-based vaccines for COVID-19 and rare cases of heart inflammation. . Myocarditis is generally the result of a viral infection, so could be unrelated to the mRNA vaccines, particularly as neither of them contain any live virus.

Express Pharma
MAY 6, 2024
Florent Bouguin, CTO of Optel Group, uncovered how ultra-low temperatures, crucial for cryopreservation, are revolutionising mRNA production for next-generation medical interventions, particularly vaccines and therapies.

Pharma in Brief
SEPTEMBER 22, 2020
On September 16, 2020, the Minister of Health signed an Interim Order Respecting the Importation, Sale and Advertising of Drugs for Use in Relation to COVID-19 (the Interim Order ) which provides new regulatory pathways to expedite the approval of COVID-19 drugs and vaccines that have not yet been approved for sale in Canada.
pharmaphorum
MAY 24, 2022
” Vaccines and drugs are available. Meanwhile, attention is now being turned to other measures to control the outbreak, including the use of vaccines against smallpox – a related virus – in a ‘ring vaccination’ approach designed to control the spread among contacts.
pharmaphorum
AUGUST 22, 2022
The EMA’s Emergency Task Force (ETF) has recommended that EU member states can use a different regimen for Bavarian Nordic’s monkeypox vaccine Imvanex/Jynneos that uses a smaller dose, to eke out limited supplies of the shot. In the study, the standard 0.5 ml dose given subcutaneously was compared to a 0.1
Pharma Packaging Solutions
JUNE 30, 2023
Vaccines: Vaccines, many of which are grown in tissue cultures, use microorganisms to provide immunity for specific diseases, and are one of the most wide-spread types of biopharma products. We offer a variety of packaging services, including vial and ampule labeling, pharmaceutical kit assembly, and more.
pharmaphorum
FEBRUARY 17, 2021
The European Commission has ordered another 200 million doses of the Pfizer/BioNTech COVID-19 vaccine Comirnaty, bringing the bloc’s total order to 500 million doses. The vaccine is based on BioNTech’s proprietary mRNA technology and was developed by both companies.

pharmaphorum
DECEMBER 11, 2020
Advisers to the FDA have voted in favour of approving Pfizer and BioNTech’s COVID-19 vaccine, with a near-unanimous backing from an expert committee. Already approved in the UK, this is the first time that mRNA technology has been used to create a vaccine. Efficacy was consistent across age, gender, race and ethnicity demographics.
pharmaphorum
MAY 30, 2022
One concern for public health experts is that with vaccination against the similar smallpox virus ending more than 40 years ago, there is little immunity in global populations to help curb the spread of infections. It says it has the capacity to produce around 30 million doses per year.

pharmaphorum
JUNE 24, 2022
Before the monkeypox outbreak, really, only the US and Canada had stockpiled our drug and very few people had actually stockpiled the vaccine between Canada, the US, and a few other countries,” Siga CEO Phil Gomez told pharmaphorum. “So I was part of the team that worked on the first SARS vaccine that went into humans.
pharmaphorum
JANUARY 13, 2022
A paper detailing the new AI has been made available on the preprint server BioRxiv and submitted to a peer-reviewed journal, said BioNTech, which alongside Pfizer developed the most widely-used mRNA-based vaccine against COVID-19. ” IHU has not yet been labelled a variant of concern by the WHO.
Pharmaceutical Technology
JULY 29, 2022
The open-label, three-cohort, multicentre trial analysed intravesical BCG, N-803 combination in BCG-unresponsive high-grade NMIBC patients. In January, ImmunityBio and Amyris have concluded a joint venture (JV) agreement announced previously to accelerate the marketing of a Covid-19 vaccine.

pharmaphorum
OCTOBER 7, 2022
Although, unlike PANORAMIC, that excluded patients who had been vaccinated against SARS-CoV-2. The UK was the first country to authorise Lagevrio, giving it a green light in November 2021 a few weeks ahead of the US, which cleared the drug with a more restrictive label. The European Commission has yet to approve it for use in the EU.

European Pharmaceutical Review
FEBRUARY 13, 2023
This is like antigens which can trigger an immune response and act as a vaccine. Firstly, using liquid chromatography and mass spectrometry, they carry out a systematic comparison of different fragments of a mRNA molecule to be tested with a similarly fragmented reference mRNA labelled with a stable carbon isotope.

Pharmaceutical Technology
JULY 14, 2022
Covid-19 vaccines stay in the spotlight. Even as regulatory agencies contemplate the authorization of Omicron-specific Covid-19 vaccines , existing versions continue to make headlines. Several companies have been charged with manufacturing different components of J&J’s vaccine.

pharmaphorum
APRIL 22, 2022
Astellas also revealed today that it is terminating the development of ASP2390, a DNA vaccine for house dust mite-induced allergic rhinitis, and GITR agonist antibody ASP1951 for cancer – both in early-stage clinical development – along with DMD gene therapies AT702, AT751 and AT753.
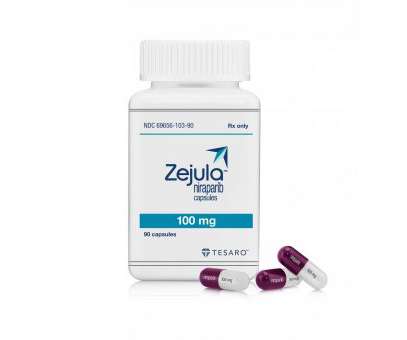
pharmaphorum
NOVEMBER 14, 2022
The narrower label for Zejula (niraparib) means it can only be used as second-line maintenance therapy after platinum-based chemotherapy in patients with these cancers whose tumours carry a germline BRCA mutation – around 15% of the population. That was due last Friday, but cancelled when GSK opted to withdraw the approval.

Pharmacy Is Right For Me
OCTOBER 21, 2022
My evenings involve music production, radio show production, podcast production, reviewing new music sent by labels/music promoters, and preparing for gigs. When the monkeypox outbreak was happening here in NYC, I used my platform to be a source of information about vaccines and TPOXX treatments.

pharmaphorum
MAY 17, 2021
Apellis Pharma has secured FDA approval for its complement C3 inhibitor Empaveli as a treatment for paroxysmal nocturnal hemoglobinuria (PNH) – with a label that will allow it to challenge Alexion’s established therapies directly. .

Pharma in Brief
AUGUST 3, 2023
The Old Guidance applied only to drugs for use in humans, whereas the Distinction Guidance now applies to drugs (both prescription and non-prescription), medical devices, natural health products, biologics, vaccines, and animal health products. Patient information.

pharmaphorum
AUGUST 27, 2020
Bioindustry association MichBio says the FDA’s labelling requirements for COVID-19 convalescent plasma (CCP) – which were published alongside the emergency use authorisation – could lead to “hundreds, if not thousands, of in-date, ready to transfuse CCP units across the country being rendered unusable.”. The @CDCgov was muzzled months ago.

Pharmaceutical Technology
JULY 22, 2022
In addition, immunity against the infection is found to wane for both previously infected individuals and the vaccinated. Acute hepatitis in previously healthy children is uncommon, despite several cases being reported every year and a few are labelled as those with unknown origin, according to experts. Username: Kai Kupferschmidt.

pharmaphorum
SEPTEMBER 21, 2020
AstraZeneca may have had a rough ride following a safety scare with its COVID-19 vaccine, but the company’s busy pipeline is producing successes in other areas. Since then AZ has changed the formulation to make it more patient-friendly and added uses in breast, ovarian, pancreatic and prostate cancer to its label.

FDA Law Blog: Biosimilars
JANUARY 3, 2024
Specifically, CVM ensures that animal drugs are safe and effective, properly made, and adequately labeled and packaged; food-producing animals only take drugs that would be safe for humans to consume; pet foods and additives are safe; and educates the public, monitors the market, and encourages development of new animal health products.

Pharma in Brief
JANUARY 19, 2023
We previously reported on the IO here.

GMPSOP
AUGUST 28, 2023
Raw materials must be inspected to confirm that the containers are intact, have been provided according to the paperwork, and have labels affixed on them identifying the raw material name, batch number, and expiry date. For example, materials with Hold, Quarantine, or Rejected labels must be kept in a quarantine location.

Pharmaceutical Technology
SEPTEMBER 16, 2022
And despite the recent pandemic that forced the industry to develop and deliver treatments and vaccines all over the world, the issue of reducing emissions has remained high on industry leaders’ agendas. The importance of sufficiently eliminating harmful greenhouse gas (GHG) emissions to limit global temperature to the 1.5-degree 1] [link].

pharmaphorum
MARCH 2, 2021
Zebra Technologies healthcare lead Chris Sullivan discusses the COVID-19 vaccine rollout and how technology can streamline the process to combat new variants. . While the rollout of the COVID-19 vaccine has signified the first signs of light at the end of the tunnel, the reality is that this public health crisis is still far from over.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content