Samsung seeks 'interchangeable' label for Humira biosimilar
pharmaphorum
AUGUST 2, 2023
Samsung seeks 'interchangeable' label for Humira biosimilar Phil.Taylor Wed, 02/08/2023 - 10:37 Bookmark this
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
pharmaphorum
AUGUST 2, 2023
Samsung seeks 'interchangeable' label for Humira biosimilar Phil.Taylor Wed, 02/08/2023 - 10:37 Bookmark this

pharmaphorum
AUGUST 25, 2023
CVS Health launches private label biosimilars unit Phil.Taylor Fri, 25/08/2023 - 08:07 Bookmark this
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Big Molecule Watch
SEPTEMBER 25, 2023
Last week, FDA released a draft guidance, “ Labeling for Biosimilar and Interchangeable Biosimilar Products ” (“2023 Draft Guidance”) that—when finalized—will revise and replace its July 2018 final guidance, “ Labeling for Biosimilar Products.”

Safe Biologics
DECEMBER 3, 2023
ASBM and GaBI Webinar Examines Policy Challenges to Interchangeable Biosimilars On November 30, ASBM and the Generics and Biosimilars Initiative (GaBI) hosted I nterchangeable Designation for Biosimilars- Ensuring Continuity of Patient Care: Upholding Interchangeability Status for Biosimilars.

Safe Biologics
NOVEMBER 12, 2023
6 “The Biosimilar Red Tape Elimination Act”, which would prevent the HHS Secretary from requiring switching studies in order for a biosimilar to be deemed “interchangeable” Under U.S. state law, only biosimilars which are interchangeable may be substituted by a pharmacist without contacting the prescriber.

Safe Biologics
DECEMBER 6, 2023
prescribers have high confidence in the safety and efficacy of biosimilars, a majority (58%) oppose third-party switching of a patient’s biologic medicine for non-medical (e.g. state law, only biosimilars which are interchangeable may be substituted by a pharmacist without contacting the prescriber. and worldwide.”
Drug Channels
OCTOBER 3, 2023
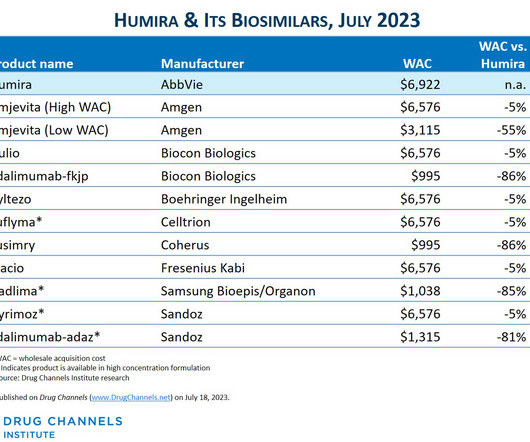
This week, I’m rerunning some popular posts while I put the finishing touches on our new 2023-24 Economic Report on Pharmaceutical Wholesalers and Specialty Distributors. CVS Health launched Cordavis, a new subsidiary that will market a private label, low-list-price version of Sandoz’ Hyrimoz. The Humira biosimilar market has arrived!

Drug Channels
DECEMBER 11, 2023
Click here to see the original post from September 2023. CVS Health has finally revealed its strategy for biosimilars of AbbVie’s Humira. Read more » Copyright © 2006-2023 Pembroke Consulting, Inc. This week, I’m rerunning some popular posts while I prepare for Friday’s Drug Channels Outlook 2024 live video webinar.

Big Molecule Watch
JANUARY 25, 2024
As we kick off 2024, we reflect on regulatory developments in the biologics and biosimilars space in 2023. Below are some of the top regulatory developments from 2023. WEZLANA is the first product to be approved as a biosimilar to STELARA.

Big Molecule Watch
AUGUST 24, 2023
In continuation of Goodwin’s previous webinar series concerning biosimilars, Big Molecule Watch is launching the 2023-2024 webinar series , which will dive deep into some of the key topics pertaining to this burgeoning industry and corresponding area of law. The post Biosimilars Webinar Series appeared first on Big Molecule Watch.

Drug Channels
SEPTEMBER 6, 2023
CVS Health has finally revealed its strategy for biosimilars of AbbVie’s Humira. Rather than announce multiple biosimilars for its pharmacy benefit manager (PBM) formulary, the company will instead launch Cordavis, a new subsidiary that will market a private label, low-list-price version of Sandoz’ Hyrimoz.

Big Molecule Watch
SEPTEMBER 7, 2023
Developing and successfully commercializing a biosimilar is a complex and expensive process. Topics that will be covered include the 271(e) Safe Harbor, peremptory challenges, labelling and timing considerations, and competitive intelligence. Click here to register for the webinar. Click here to register for the webinar.

Big Molecule Watch
NOVEMBER 21, 2023
District Court for the District of New Jersey, alleging infringement of 15 patents under the BPCIA based on DRL’s submission of an aBLA for DRL_RI, a proposed biosimilar of RITUXAN (rituximab) and DRL’s provision of Notice of Commercial Marketing with respect to the same. Stay tuned to Big Molecule Watch for more updates on this litigation.

FDA Law Blog: Biosimilars
AUGUST 21, 2023
Livornese — I saw the sign…and the answer is no—FDA-approved labeling apparently is not enough under state failure-to-warn laws, according to certain courts. A recent state law failure-to-warn case in the SDNY makes that very point.

FDA Law Blog: Biosimilars
AUGUST 17, 2023
Tobolowsky — In January 2023, Vanda Pharmaceuticals, Inc. FDA-2023-P-0313 and FDA-2023-P-0344 ) regarding its product Hetlioz (tasimelteon). a)(8)(iv), interpret these provisions to also allow changes due to an aspect of labeling protected by patent or exclusivity. 355(j)(2)(A)(v). 355(j)(2)(A)(v).

FDA Law Blog: Biosimilars
MAY 23, 2023
Food and Drug Administration (FDA) released a draft update to its Compliance Policy Guide (CPG) for FDA staff on the Agency’s enforcement of major food allergen labeling and cross-contact. The draft CPG directs FDA field staff to examine possible food product adulteration due to labeling related to allergen cross-contact.

PharmaShots
FEBRUARY 3, 2023
4D Molecular Therapeutics Receives the US FDA’s IND Clearance of 4D-150 for the Treatment of Diabetic Macular Edema Date: Feb 03, 2023 | Tags: 4D Molecular Therapeutics, 4D-150, Diabetic Macular Edema, Regulatory, US, FDA, IND AstraZeneca and Amgen Receive the US FDA’s Approval of Tezspire (tezepelumab) for the Treatment of Severe Asthma (..)

FDA Law Blog: Biosimilars
MARCH 29, 2023
Richardson — Early on March 29, 2023, FDA announced the landmark approval of Narcan (naloxone hydrochloride) Nasal Spray for use as a nonprescription opioid overdose reversal agent. According to this announcement, FDA approval of RiVive is anticipated in July 2023 and the U.S. By Kalie E. launch would be in early 2024.

Big Molecule Watch
MARCH 27, 2023
On March 20, 2023, Coya Therapeutics, Inc. (“ Coya ”) announced a worldwide agreement with Dr. Reddy’s Laboratories Ltd. (“Dr. Reddy’s”) to license Dr. Reddy’s proposed biosimilar abatacept for the development and commercialization of COYA 302 for the treatment of neurodegenerative conditions.

PharmaShots
MARCH 17, 2023
5-Adapted Bivalent Booster to Treat COVID-19 in Children ≤5 Years Date: Mar 15, 2023 | Tags: Pfizer, BioNTech, Omicron BA.4/BA.5-Adapted 5-Adapted Bivalent Booster to Treat COVID-19 in Children ≤5 Years Date: Mar 15, 2023 | Tags: Pfizer, BioNTech, Omicron BA.4/BA.5-Adapted

European Pharmaceutical Review
DECEMBER 20, 2023
In Q1 2023, the European Commission (EC) announced proposed changes to EU pharmaceutical legislation. Changes to regulatory data protection periods are of particular interest to biopharma” Changes to regulatory data protection (RDP) periods are of particular interest to biopharma (originators and generic / biosimilar manufacturers).

Big Molecule Watch
JUNE 29, 2023
As we previously reported , Biogen sued Sandoz and Polpharma (“Defendants”) in a BPCIA litigation related to Defendants’ natalizumab biosimilar. Oral argument was held on May 17, 2023. On June 27, 2023, the Court filed a redacted version of its order.

PharmaShots
MAY 3, 2023
Our team at PharmaShots has summarized 15 key events of the biosimilar space of April 2023 1. results from the P-III clinical trial of CT-P42, a biosimilar referencing Eylea. results from the P-III clinical trial of CT-P42, a biosimilar referencing Eylea. Ximluci is supplied as a 2.3mg/0.23 mL single-use vial inj.

PharmaShots
MARCH 1, 2023
In preclinical studies, HLX15 is highly similar to daratumumab Amgen Launches First Humira (adalimumab) Biosimilar Amjevita in the US Date- February 01, 2023 Product: Amjevita The company has launched Amjevita (adalimumab), the first biosimilar referencing blockbuster drug Humira on the US market for certain serious inflammatory diseases.

FDA Law Blog: Biosimilars
JUNE 11, 2023
Clissold — In May 2023, CDER and CBER published a draft guidance titled “Pediatric Drug Development: Regulatory Considerations – Complying with the Pediatric Research Equity Act [“PREA”] and Qualifying for Pediatric Exclusivity Under the Best Pharmaceuticals for Children Act [“BPCA”]” (the “Draft Guidance”). Tobolowsky & David B.

FDA Law Blog: Biosimilars
AUGUST 4, 2023
DASCA amended the CSA by revising and adding specified substances to the definition of “anabolic steroid,” providing for the temporary and permanent scheduling of anabolic steroids and adding labeling requirements for products containing anabolic steroids. Implementation of the Designer Anabolic Steroid Control Act of 2014 , 88 Fed.

PharmaShots
APRIL 5, 2023
neovascular (wet) AMD Additionally, the availability of Byooviz could enable more patients to access this type of vision-saving therapy as it brings a sustainable and cost-effective anti-VEGF treatment to Canadian vision care Byooviz was approved in Canada for the treatment of multiple eye disorders.

FDA Law Blog: Biosimilars
APRIL 25, 2023
This provision became effective as of March 29, 2023. It will become part of the “refuse to accept” (RTA) checklist on October 1, 2023. Timeline Section 524B became effective on March 29, 2023. Until this deadline, FDA generally intends not to issue RTA decisions based solely on the information required by section 524B.

FDA Law Blog: Biosimilars
JANUARY 1, 2024
Maybe that’s also why FDA last week publicized the highest number of important Warning Letters of the year (compared with prior releases in 2023). Perhaps FDA wanted us to remember 2023 as the year FDA succeeded in uncovering critical defects in drug and device manufacturing, and in critical trials. Terragene S.A., Terragene S.A.,

FDA Law Blog: Biosimilars
MAY 15, 2023
States frequently review labels (and labeling) for animal food products. The CFI includes a list of common foods that “may be appropriate for use in animal food and serve as a tool for use during review of ingredients on an animal food label.” Part of this review involves the status of the ingredients included in the product.

FDA Law Blog: Biosimilars
JUNE 22, 2023
FDA hailed its authorization of the Lucira OTC home test for influenza and COVID-19 as “underscor[ing] the Agency’s continued commitment to increase availability of accurate and reliable at-home diagnostic tests” ( April 2023 FDA Update to CDC ). This usually requires human factors or usability testing and labeling comprehension testing.

FDA Law Blog: Biosimilars
MAY 19, 2023
Serial testing was defined as testing symptomatic individuals twice over three days with at least 48 hours between tests, which is in accordance with study findings from the National Institute of Health referenced here and current EUA labeling for serial testing. Customer Support Help line).

FDA Law Blog: Biosimilars
OCTOBER 24, 2023
Mullen — As of October 1, 2023, all 510(k) submissions, unless exempted, must be submitted to FDA using the electronic Submission Template And Resource ( eSTAR ). On September 29, 2023, FDA released a draft guidance on Electronic Submission Template for Medical Device De Novo Requests. By Philip Won & Allyson B.

FDA Law Blog: Biosimilars
JANUARY 3, 2024
Specifically, CVM ensures that animal drugs are safe and effective, properly made, and adequately labeled and packaged; food-producing animals only take drugs that would be safe for humans to consume; pet foods and additives are safe; and educates the public, monitors the market, and encourages development of new animal health products.

Big Molecule Watch
DECEMBER 28, 2023
According to PWC , “2023 was a reasonably strong year for the pharmaceutical and life sciences sector with both deal value and volume of M&A close to pre-pandemic levels.” Below is a recap of the top M&A deals in 2023 that focus on biologics and biosimilars and were covered on the Big Molecule Watch.
Quality Matters
APRIL 20, 2023
Quality across the product lifecycle The role of standards goes far beyond the drug manufacturing process – from the sourcing of raw materials to packaging, labeling, distribution, and point-of-care administration – to help ensure quality across the pharmaceutical product lifecycle. kristine.gates… Thu, 04/20/2023 - 08:51 Supply Chain

FDA Law Blog: Biosimilars
JUNE 27, 2023
On June 12, 2023, FDA issued a public notice to solicit comments on the information collection related to the voluntary submission of allegations of regulatory misconduct to CDRH. Any comments to the public notice must be submitted by August 11, 2023. We hope the status is more than just an “Under Review” label.

FDA Law Blog: Biosimilars
JUNE 28, 2023
By Riëtte van Laack — On June 8, 2023, bipartisan legislation creating a new category of animal feed ingredients, named zootechnical animal food substances, was introduced. On June 15, 2023, an amendment modeled after the Innovative FEED Act, was proposed as an amendment to S.1844

FDA Law Blog: Biosimilars
MAY 8, 2023
FDA recommends manufacturers submit each modification, associated labeling changes and specific performance evaluation in the Modification Protocol (MP). Update procedures : The MP should describe how the manufacturer will update the software and any associated labeling that will be required for the modifications.

FDA Law Blog: Biosimilars
OCTOBER 10, 2023
Labeling and Implant ID Card Lastly, it is critical that patients are provided with implant information as it pertains to their devices. separate patient labeling). Manufacturers should create user-friendly instructions for use that facilitate patient understanding of potential risks over the expected lifespan of the implant.

PharmaShots
APRIL 3, 2023
Shots: The first quarter of 2023 highlights major acquisitions in the pharma and biotech industry along with multiple approvals. Date - Jan 04, 2023 Product – N/A The acquisition is completed from a shareholder group led by Nordic Capital, in an all-cash transaction valued at $2.8B per ADS up front & CVR of ~$2.50

FDA Law Blog: Biosimilars
MARCH 27, 2023
Such codes need to be placed on device labels and packages to allow devices to be easily identified and tracked throughout their lifecycle, except where the rule provided for an exception or alternative. So, what is special about September 24, 2023? Devices can have both a UPC code and a UDI on their label and package.

FDA Law Blog: Biosimilars
APRIL 30, 2023
1067, the “ Ensuring Timely Access to Generics Act of 2023 ,” and it would fundamentally transform the playing field for NDA, ANDA, BLA, and aBLA applicants seeking to preserve their rights in the wake of an adverse FDA approval decision. That bill is S.

Safe Biologics
MAY 10, 2024
Biden Administration HHS Budget Would Permit Third-Party Substitution of All Biosimilars On March 11th, the Biden Administration released its FY25 HHS Budget. and Europe alike, prescribers can already substitute any biosimilar for its reference product. Currently there are 10 biosimilars that can be substituted by U.S.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content