FDA Updates Draft Guidance on Promotional Communications of Prescription Biologics and Biosimilars
Big Molecule Watch
MAY 3, 2024
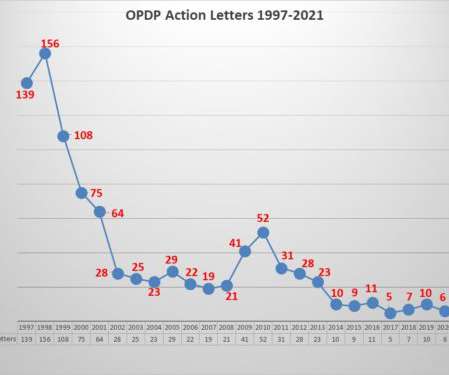
The updated guidance includes information on the general requirements for the content of FDA-regulated promotional communications about reference or biosimilar products and provides some examples to illustrate the FDA’s current recommendations for addressing reference, biosimilar, and interchangeable biosimilar products in product promotion.

































Let's personalize your content