What are the elements of quality control process in pharmaceuticals
GMPSOP
OCTOBER 28, 2023
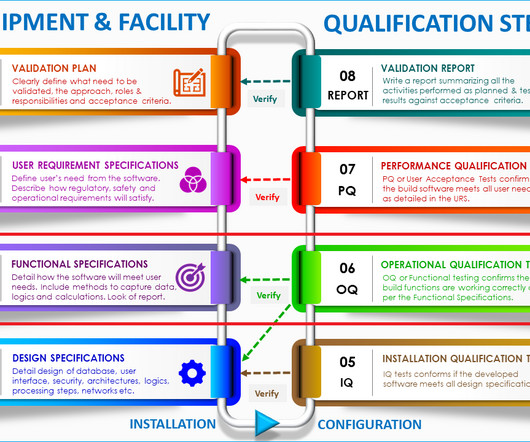
Typical flow of samples in a quality control laboratory The quality control laboratory receives samples that are collected randomly at the beginning, middle, and end of manufacturing processes. . – Record or capture all generated raw data directly, promptly, and legibly. Typical documentation in a quality control process?








Let's personalize your content