New blood test can identify presence of cancer
Pharma Times
JANUARY 5, 2022
The test is a technological first in determining the metastatic status of a cancer without using prior insight of the primary cancer presented.
Pharma Times
JANUARY 5, 2022
The test is a technological first in determining the metastatic status of a cancer without using prior insight of the primary cancer presented.

pharmaphorum
JANUARY 9, 2022
US medical device company Abbott has said it plans to launch a new range of ‘biowearable’ health devices aimed at consumers, in a move that builds on its expertise in glucose monitoring. The company’s chief executive – Robert Ford – made the announcement at the massive CES tech trade show in Las Vegas, a venue usually dedicated to new TVs, gaming equipment, new smartphones and the like.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Outsourcing Pharma
JANUARY 12, 2022
The company plans to launch a Phase Ib/II study of its compound NVG-291 in collaboration with researchers at Northwestern Universityâs Shirley Ryan AbilityLab.
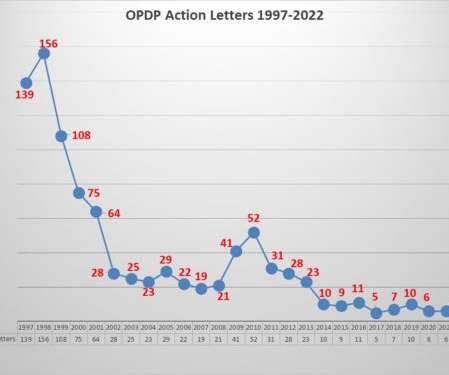
Eye on FDA
JANUARY 26, 2022
FDA’s Office of Prescription Drug Promotion (OPDP) issued the first regulatory action letter for 2022. This one has some notable characteristics. As has been frequently noted, enforcement actions by OPDP have diminished considerably over the years. Another characteristic of recent enforcement actions over the past several years is that smaller, less well-known companies have mostly been the recipients of letters from OPDP.

Speaker: Chris Antypas and Josh Halladay
Access to limited distribution drugs and payer contracts are key to pharmacy expansion. But how do you prepare your operations to take the next step? Meaningful data: Collect and share clinical data regarding outcomes, utilization, and more Reporting: Limited distribution models require efficient tracking and reporting systems Workflows: Align workflows with specific pharma and payer contractual requirements For in-depth, expert insights on pharmacy expansion, watch this webinar from Inovalon.
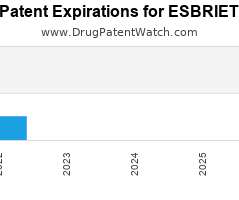
Drug Patent Watch
JANUARY 24, 2022
Annual Drug Patent Expirations for ESBRIET Esbriet is a drug marketed by Genentech Inc and is included in two NDAs. It is available from one supplier. There are twenty-one patents…. The post New patent expiration for Genentech Inc drug ESBRIET appeared first on DrugPatentWatch - Make Better Decisions.

NY Times
JANUARY 14, 2022
A federal judge found that Mr. Shkreli violated state and federal antitrust laws by trying to maintain a monopoly over a lifesaving drug and must pay back the excess profits.
Pharmacist Digest brings together the best content for pharmacists from the widest variety of industry thought leaders.

pharmaphorum
JANUARY 27, 2022
Walter Greenleaf, PhD, neuroscientist and digital health expert at Stanford University’s Virtual Human Interaction Lab, tells us about the new book Applied Virtual Reality in Healthcare: Case Studies and Perspectives and why he and his co-authors gathered insights from some of the most prominent figures in the field of medical extended reality (XR).
Outsourcing Pharma
JANUARY 10, 2022
Joining with a noted research foundation, the group is aiming for earlier detection and treatment of the disease, which hits nearly 500K people annually.

Pharma Marketing Network
JANUARY 19, 2022
Talk to me. Pharma brands that want deep engagements need to look at and listen to social audio. It’s not surprising. In fact, it’s the most direct route to your goal. You want to talk to doctors? To start the conversation, we put pictures in front of them, bombarded them with videos, retarget them, and then send them emails. We want to talk, and they want to listen.

Drug Patent Watch
JANUARY 21, 2022
Annual Drug Patent Expirations for NAPROXEN+SODIUM Naproxen Sodium is a drug marketed by Bionpharma Inc, Catalent, Puracap Pharm Llc, Actavis Labs Fl Inc, Able, Amneal Pharms Ny, Aurobindo Pharma Ltd,…. The post New patent for Bionpharma Inc drug NAPROXEN SODIUM appeared first on DrugPatentWatch - Make Better Decisions.

Advertisement
Are you still using workarounds to manage your daily operations? To achieve peak performance, it's time to explore other options for specialty and infusion pharmacy software. Streamline pharmacy operations and improve clinical performance with automated processing, real-time data exchange, and electronic decision support. Download this helpful infographic to: Drive efficiency and patient adherence from referral receipt to delivery and ongoing care – all with our Pharmacy Cloud.
NY Times
JANUARY 26, 2022
Myocarditis, or inflammation of the heart muscle, occurred in 1 of 12,361 boys between 12 and 15 years old within a week of receiving a second dose of the Pfizer-BioNTech vaccine, researchers found.

Pharma Times
JANUARY 13, 2022
AstraZeneca (AZ) has reported preliminary data from a trial it conducted on its COVID-19 booster shot, Vaxzevria.

pharmaphorum
JANUARY 19, 2022
Just months after the venture was unveiled, controversial biotechnology company Altos Labs has officially launched with $3 billion in funding secured from investors. Dubbed an ‘anti-ageing’ company, Altos has gained mainstream attention for its focus on ‘rejuvenation’ and biological reprogramming technology. While the specific research goals have been kept a closely guarded secret, some scientists propose that rejuvenating cells in a lab could be a path to prolonging huma

Outsourcing Pharma
JANUARY 18, 2022
Regulatory bodies in the European Union have launched an initiative to develop the region as a focal point for clinical research and to better integrate clinical research in the European health system.

Drug Channels
JANUARY 19, 2022
For 2022, the three largest pharmacy benefit managers (PBMs)—Caremark (CVS Health), Express Scripts (Cigna), and OptumRx (United Health Group)— increased the number of drugs they excluded from their standard formularies. Each exclusion list contains 400 to 500 products. Growth in the number of excluded drugs slowed, due partly to the fact that so many drugs have already been dropped from PBMs’ formularies.

Pharma Marketing Network
JANUARY 27, 2022
Tune in to the Pharma Marketing Network and industry experts Novartis, UCB, GSK and others, for a fireside talk on hot topics around what has changed in the last year and how to align your pharma marketing strategies accordingly. Three Panels: . Engaging Oncologists in 2022. Emerging Digital Channels and Navigating the Social Landscape: What’s Hot, What’s Not, What’s the Next Big Thing.

NY Times
JANUARY 11, 2022
A former Africa correspondent, who covered the height of the AIDS epidemic there 15 years ago, finds lessons in the remarkable progress against that virus for our current fight.

Pharma Times
JANUARY 21, 2022
Psyros Diagnostics has signed an agreement to be acquired by the Swedish company, Prolight Diagnostics AB. Psyros’s ground-breaking Point-of-Care (POC) technology enables the measurement of disease-indicating molecules in extremely low concentrations–in ten minutes or less.
pharmaphorum
JANUARY 11, 2022
With evidence growing that the current crop of COVID-19 vaccines are less protective against infection with Omicron, both Pfizer and Moderna have announced plans to develop new versions of their mRNA-based shots that are tailored to the new variant. Pfizer has said that a COVID-19 jab specifically targeting Omicron, as well as other existing variants, will begin human trials later this month and could be available as early as March.

Outsourcing Pharma
JANUARY 13, 2022
With the amount of available data increasing and technology evolving, says a company leader, the job of data manager is more demanding than ever before.

Drug Channels
JANUARY 10, 2022
PCMA Business Forum. February 28 – March 1, 2022 JW Marriott Grande Lakes | Orlando, FL Register Today! www.pcma.com/businessforum. The PCMA Business Forum is PCMA's largest business networking and educational conference. PCMA’s PBM Members send a large number of executives from relevant business units including pharma and trade relations, specialty pharmacy, clinical and patient care services, and the c-suite.

Eye on FDA
JANUARY 5, 2022
FDA has scheduled the first advisory committee meetings of 2022. One meeting of the Oncologic Drugs Advisory Committee is set for February 10 to discuss a new application for the proposed treatment of Non-Small Cell Lung Cancer (NSCLC. And a meeting of the Anesthetic and Analgesic Drug Products Advisory Committee is meeting jointly with the Drug Safety and Risk Management Advisory Committee on February 15 to consider an NDA in the pain category.

NY Times
JANUARY 17, 2022
For months, the jail’s doctor has promoted the drug, which health experts say should not be used to treat or prevent Covid-19.

Pharma Times
JANUARY 4, 2022
Pfizer’s Paxlovid has a success rate of nearly 90% as a prophylaxis treatment against severe COVID-19 when administered shortly after the patient has been infected with the virus.

pharmaphorum
JANUARY 14, 2022
The European Commission, EMA and national regulators within the EU have launched an initiative to change the way clinical trials are designed and run in order to position the bloc as an international “focal point” for clinical research. The broad strokes of the proposals are laid out in a document entitled Accelerating Clinical Trials in the EU (ACT EU) that lists priority actions that will be taken, including measures to allow greater use of innovative clinical trial designs.

Outsourcing Pharma
JANUARY 20, 2022
An expert from patient solutions firm Trialbee discusses how the social media firm's recent ban on targeted advertising might impact patient recruitment.

Drug Channels
JANUARY 18, 2022
Join Dr. Adam J. Fein, CEO of Drug Channels Institute (DCI), for three live video webinars during 2022. These live, interactive events will be broadcast via Zoom from the Drug Channels Video studio in beautiful downtown Philadelphia. Click here to order the Drug Channels 2022 Video Webinar Series. During these events, Dr. Fein will address the latest issues confronting the U.S. drug channel.

Eye on FDA
JANUARY 12, 2022
Each year we look back to take stock of how the agency has been communicating in the course of the previous year and assess how that might compare to years gone by. In fact from year to year, there is a good deal of change that occurs. When Dr. Scott Gottlieb took the reins of FDA as Commissioner, the amount of communications material put out jumped enormously.

NY Times
JANUARY 14, 2022
After one of his sons fell into a deep depression, he helped him recover. He later licensed an antidepressant that was a boon to his pharmaceutical company.

Pharma Times
JANUARY 11, 2022
There is currently no cure for cystic fibrosis (CF), a debilitating, progressive condition with over 10,830 people in the UK currently diagnosed with the disease.

pharmaphorum
JANUARY 12, 2022
Roche has launched what it says is a first-of-its kind handheld glucose management device, backed up by a digital platform that aims to simplify the work of doctors and nurses as they deliver care to patients. The cobas pulse system is a point-of-care device that combines a glucose test strip reader with a touch screen that is used for displaying patient information and data, as well as analysing and sharing clinical results.

Outsourcing Pharma
JANUARY 24, 2022
The CRO's work with La Jolla Institute for Immunology, centered on a unique mouse model, is intended to increase understanding of how the COVID-19 virus operates.

Drug Channels
JANUARY 21, 2022
Today’s guest post comes from Chris Dowd, Senior Vice President of Market Development at ConnectiveRx. Chris discusses how COVID-19 is impacting stakeholders across the healthcare system, including patients, providers, pharmacies, and manufacturers. On February 22nd at 1:00 pm ET, ConnectiveRx will host Four Big Implications for Patients and the Prescription Journey in 2022 , a free webinar about the pandemic’s impact on key healthcare stakeholders.
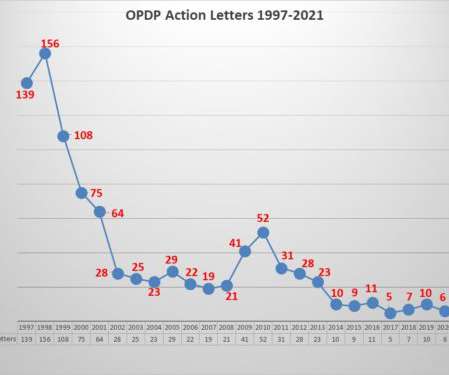
Eye on FDA
JANUARY 9, 2022
Once again it was a quiet year with respect to the FDA’s Office of Prescription Drug Programs (OPDP). As noted in the past, there are two primary means for understanding the agency’s latest thinking with respect to promotional communications from pharmaceutical companies – the content and pattern of enforcement and the issuance of guidance documents to shed light on the parameters that exist.
NY Times
JANUARY 11, 2022
If the preliminary decision is finalized this spring, it would sharply limit the number of patients who use the expensive drug.
Let's personalize your content