'Nobel Prize of pharma' helps put Huma's digital health platform on the global map
PharmaVoice
JUNE 23, 2022
The company’s CEO explains how its Prix Galien award-winning app bridges patient care, treatment support and clinical research.

PharmaVoice
JUNE 23, 2022
The company’s CEO explains how its Prix Galien award-winning app bridges patient care, treatment support and clinical research.

European Pharmaceutical Review
JUNE 24, 2022
The Greek state has filed a lawsuit against Novartis , asking for € 214 million in compensation for non-pecuniary damages* the country may have suffered due to alleged illegal payments to government officials and healthcare providers. Commenting on the lawsuit , Greece’s Health Minister Thanos Plevris, stated that the country is seeking compensation for the damage it has suffered “from the actions that Novartis itself has admitted to in the USA that concerns payments to doctors.”.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
pharmaphorum
JUNE 23, 2022
Eisai’s digital tool to allow self-assessment of cognitive performance – NouKNOW – will be used in a Japanese project aimed at improving the management of dementia. The smartphone app – which uses a cognition-checking algorithm developed by Australian digital health company Cogstate – will be deployed in Bunkyo City, Tokyo in a dementia monitoring programme.

PharmExec
JUNE 20, 2022
The world’s largest cancer research meeting, the American Society of Clinical Oncology (ASCO) Annual Meeting, recently convened in person in Chicago after two years of being held virtually. Sara Baker and Kristine Austria Fan of Marina Maher Communications provide their takeaways from the meeting.

Speaker: Chris Antypas and Josh Halladay
Access to limited distribution drugs and payer contracts are key to pharmacy expansion. But how do you prepare your operations to take the next step? Meaningful data: Collect and share clinical data regarding outcomes, utilization, and more Reporting: Limited distribution models require efficient tracking and reporting systems Workflows: Align workflows with specific pharma and payer contractual requirements For in-depth, expert insights on pharmacy expansion, watch this webinar from Inovalon.

PharmaVoice
JUNE 20, 2022
In the evolution of healthcare, data is the linchpin to making drug development more efficient.

Pharmaceutical Technology
JUNE 24, 2022
As regulatory agencies gear up for another round of Covid-19 vaccine deliberations centered on emerging variants, Moderna has released data on its booster’s efficacy against Omicron subvariants. In addition to providing protection against the Omicron BA.1 variant, on 22 June, Moderna said its booster also showed a “potent” antibody response against Omicron subvariants BA.4 and BA.5.
Pharmacist Digest brings together the best content for pharmacists from the widest variety of industry thought leaders.

Pharma Times
JUNE 22, 2022
Novartis receives approval from the European Commission for Tabrecta for the treatment of non-small cell lung cancer

PharmaVoice
JUNE 23, 2022
Even execs get the blues — and when they’re ready for a fresh start, Leslie Loveless, CEO of Slone Partners, says these dos and don’ts could make or break their next job move.

Pharmaceutical Technology
JUNE 23, 2022
The European Commission (EC) has approved Novartis’ Tabrecta (capmatinib) as a monotherapy to treat advanced non-small cell lung cancer (NSCLC) in adults. The treatment is intended for patients with NSCLC harbouring alterations leading to mesenchymal-epithelial-transition factor gene (MET) exon 14 (METex14) skipping and for those who need systemic therapy after previous treatment with immunotherapy and/or platinum-based chemotherapy.

pharmaphorum
JUNE 21, 2022
Armed with new phase 3 data, AstraZeneca is preparing to file for regulatory approval of eplontersen, an antisense drug for one of the complications of the disease transthyretin amyloidosis (ATTR) it licensed from Ionis in a $3.6 billion deal last year. Eplontersen – previously known as IONIS-TTR-LRX – is designed to switch off the production of transthyretin (TTR), a protein which builds up in the disease to toxic levels causing heart problems (cardiomyopathy) and nerve damage (polyneuropathy).

Advertisement
Are you still using workarounds to manage your daily operations? To achieve peak performance, it's time to explore other options for specialty and infusion pharmacy software. Streamline pharmacy operations and improve clinical performance with automated processing, real-time data exchange, and electronic decision support. Download this helpful infographic to: Drive efficiency and patient adherence from referral receipt to delivery and ongoing care – all with our Pharmacy Cloud.

European Pharmaceutical Review
JUNE 24, 2022
2022 is probably the biggest year in EU pharmaceutical law that I have seen in 30 years. It is an exciting year… but whether the changes will be a net positive or a net negative is yet to be seen” Maarten Meulenbelt, Partner, Sidley Austin LLP. 2022 is undeniably a critical year for change in the pharmaceutical industry: for the first time in several decades, the European Union (EU)’s pharmaceutical legislation is up for review.

PharmaVoice
JUNE 21, 2022
Deal-maker, mentor and biotech industry mover and shaker, the COO and general counsel brings her 'smiling tiger' skills to her new role at the emerging company.

Pharmaceutical Technology
JUNE 24, 2022
Novavax has obtained emergency use authorization (EUA) for its Covid-19 vaccine, Nuvaxovid (NVX-CoV2373), from the Taiwan Food and Drug Administration for use in people of the age 18 years and above. The protein-based vaccine is engineered from the genetic sequence of the SARS-CoV-2 virus’ initial strain. Developed utilising Novavax’s recombinant nanoparticle technology to generate antigen obtained from the coronavirus spike (S) protein, the vaccine is also formulated with the company’s saponin-

pharmaphorum
JUNE 20, 2022
Speculation that Merck & Co may be preparing a $30 billion takeover bid for Seagen – bolstering its oncology ambitions – has the biopharma community aflutter. The rumour was first published in the Wall Street Journal, which suggests that a takeover is just one option on the table, along with a marketing deal that would bind the two companies together without the risk of antitrust issues holding up or blocking a merger.

Pharma Times
JUNE 20, 2022
The discovery may lead to development of drugs that halt disease progression and stop memory loss

PharmaVoice
JUNE 21, 2022
In her decades-long career, Mary Pittman, CEO of the Public Health Institute, has seen — and helped solve — one public health mystery after another.
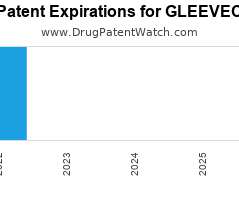
Drug Patent Watch
JUNE 18, 2022
Annual Drug Patent Expirations for GLEEVEC Gleevec is a drug marketed by Novartis and is included in two NDAs. It is available from one supplier. There is one patent protecting…. The post New patent expiration for Novartis drug GLEEVEC appeared first on DrugPatentWatch - Make Better Decisions.

pharmaphorum
JUNE 22, 2022
Pfizer has harnessed the expertise of health data specialist Truveta to provide close monitoring of safety and other signals for its products, including COVID-19 vaccine Comirnaty, almost in real-time. Truveta’s platform is based on electronic medical record data – updated daily – for more than 50 million people in the US, harvesting anonymised patient care information from tens of thousands of clinical care sites across 42 states.
Pharma Times
JUNE 21, 2022
Pre-clinical screening technology could increase cancer test numbers by 50 times

PharmaVoice
JUNE 21, 2022
Regulatory experts weigh in on ways to enhance the controversial drug approval approach.
Outsourcing Pharma
JUNE 22, 2022
The company reports it has met the enrollment target for a trial of its sonic beam therapy, one step closer to submitting for FDA approval of the product.

pharmaphorum
JUNE 22, 2022
The pandemic highlighted how fragile the global manufacturing network is, as supply of certain products was limited and regulatory inspections became difficult to carry out. Ben Hargreaves finds out how the issue of foreign manufacturing has become a major US political talking point and how this could influence the pressures facing the industry. In a global economy, across a number of industries, low-margin products are often manufactured in countries where labour is cheap.

Pharma Times
JUNE 23, 2022
Global surveillance systems show that recorded cases do not include any deaths

Drug Channels
JUNE 21, 2022
National Association of Specialty Pharmacy (NASP) 2022 Annual Meeting & Expo. September 19-22, 2022 | Gaylord Palms Resort & Convention Center | Orlando, FL. www.naspnet.org/annualmeeting. The National Association of Specialty Pharmacy (NASP) will host the NASP 2022 Annual Meeting & Expo September 19-22. NASP will celebrate their 10th Anniversary and welcome attendees to a new conference venue – the gorgeous Gaylord Palms Resort & Convention Center, located in Orlando, Florida.

European Pharmaceutical Review
JUNE 24, 2022
BioNTech has initiated construction of its first vaccine production facility in Africa. Located in Kigali, Rwanda, the mRNA manufacturing facility will be the first of three planned sites across the continent, with further factories to come in Senegal and South Africa. Together this network of factories will supply therapies and vaccines solely for people residing in member states of the African Union.

pharmaphorum
JUNE 20, 2022
Pfizer has acquired an 8.1% stake in French vaccines developer Valneva at a cost of €90.5 million (around $95 million) and rejigged the terms of their alliance to develop a shot for Lyme disease. The deal gives Valneva a cash injection that it will be able to use to advance Lyme disease candidate VLA15 towards the market, but the revised licensing terms means that the French biotech will shoulder more of the financial cost of the development programme.
Pharma Times
JUNE 23, 2022
The company has renewed its pledge to eliminate neglected tropical diseases and malaria over five years

Drug Channels
JUNE 24, 2022
Today’s guest post comes from Logan Melchione, Senior Director of Account Management at Paysign. Logan describes how some patient affordability vendors capture funds that were intended for patients. She then provides manufacturers with ideas to better manage and oversee copay, bridge, and voucher programs. To learn more, register for Paysign’s July 13 webinar: Affording Patient Affordability Programs: The Truth About Voucher and Bridge Pricing.

Outsourcing Pharma
JUNE 21, 2022
If new restrictions are placed on the FDAâs Accelerated Approval pathway, as many as two thirds of the treatments that use this pathway would never reach patients, according to research released at the BIO International Convention last week.
pharmaphorum
JUNE 21, 2022
While proponents argue waiving IP on COVID-19 vaccines would better enable low- and middle-income countries to inoculate their populations, the pharma industry has called the proposals “unnecessary and harmful to innovation”. Proposals to waive COVID-19 product patents have been described as “political posturing” and an “answer to a problem that does not exist”.

Pharma Times
JUNE 24, 2022
MHRA extends UCB’s therapy for the treatment of partial-onset seizures
Pharmaceutical Technology
JUNE 24, 2022
invoX Pharma has signed a definitive agreement for the acquisition of all issued and outstanding shares of clinical-stage biopharma firm F-star Therapeutics, in a deal valued at nearly $161m or $7.12 for each share in cash. invoX Pharma is a fully owned Sino Biopharmaceutical subsidiary. The latest deal will expedite the strategy of invoX to develop the International Biopharmaceutical R&D Platform of Sino Biopharm outside of China to transform the lives of patients globally.

PharmExec
JUNE 20, 2022
Additions of Andrew Johnson and Bridget Seay strengthen epocrates’ evolution as a commercial organization with best-in-class market intelligence and products.

pharmaphorum
JUNE 23, 2022
Novartis has shouldered its way into the in vivo gene editing category via a deal with US biotech Precision BioSciences, focused on a therapy for sickle cell disease (SCD). The Swiss pharma group is paying $75 million upfronting cash and equity to get the ball rolling on the collaboration, with another $1.4 billion in potential milestones if the project advances through development and onto the market.

Pharma Times
JUNE 20, 2022
The joint aim of the companies was to provide vaccines to vulnerable communities
Let's personalize your content