NHS launches 40 ‘long COVID’ clinics
Pharma Times
NOVEMBER 16, 2020
It is hoped that the move will help to tackle the persistent symptoms – such as fatigue and brain fog – suffered by people who have developed the condition
Pharma Times
NOVEMBER 16, 2020
It is hoped that the move will help to tackle the persistent symptoms – such as fatigue and brain fog – suffered by people who have developed the condition
NY Times
NOVEMBER 19, 2020
In a review of several trials, the World Health Organization found that Gilead’s drug did not improve survival rates for patients nor did it help them recover.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

pharmaphorum
NOVEMBER 20, 2020
Doctors in the US are to get another option to treat COVID-19 after the FDA granted an Emergency Use Authorization (EUA) for Eli Lilly’s baricitinib. Baricitinib is the active ingredient in Lilly’s rheumatoid arthritis drug Olumiant and is the latest example of a drug being repurposed to combat COVID-19. Most of the drugs attempt to tackle the inflammation that can cause complications in patients with severe disease.
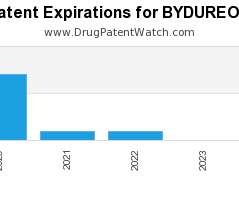
Drug Patent Watch
NOVEMBER 15, 2020
Annual Drug Patent Expirations for BYDUREON Bydureon is a drug marketed by Astrazeneca Ab and is included in two NDAs. It is available from one supplier. There are thirty patents…. The post New patent for Astrazeneca Ab drug BYDUREON appeared first on DrugPatentWatch - Make Better Decisions.

Speaker: Chris Antypas and Josh Halladay
Access to limited distribution drugs and payer contracts are key to pharmacy expansion. But how do you prepare your operations to take the next step? Meaningful data: Collect and share clinical data regarding outcomes, utilization, and more Reporting: Limited distribution models require efficient tracking and reporting systems Workflows: Align workflows with specific pharma and payer contractual requirements For in-depth, expert insights on pharmacy expansion, watch this webinar from Inovalon.

Pharma Times
NOVEMBER 16, 2020
The funds will help expand the scale of SARS-CoV-2 virus sequencing in the hope of revealing new opportunities for intervention
NY Times
NOVEMBER 18, 2020
The company said it planned to apply for emergency approval from the Food and Drug Administration “within days.
Pharmacist Digest brings together the best content for pharmacists from the widest variety of industry thought leaders.

Outsourcing Pharma
NOVEMBER 18, 2020
The federal agency has offered updates, advice and action regarding the COVID-19 pandemic, helping life-sciences professionals stay on top of developments.

Pharma Times
NOVEMBER 16, 2020
The recommendations include new treatments for cancer and intraocular pressure and prevention of uncomplicated flu.

NY Times
NOVEMBER 16, 2020
Moderna is the second company to report preliminary results from a large trial testing a vaccine. But there are still months to go before it will be widely available to the public.

pharmaphorum
NOVEMBER 20, 2020
COVID-19 has turned the world on its head and healthcare systems have had to respond rapidly to match the sudden changes created by lockdown. Digital health had already been building a presence before the pandemic, but the tools it offers have been essential to counter the disruption caused by the coronavirus, reports Richard Staines. COVID-19 isn’t going away any time soon but when, and if, this crisis subsides the healthcare systems that are left behind will have changed radically.

Advertisement
Are you still using workarounds to manage your daily operations? To achieve peak performance, it's time to explore other options for specialty and infusion pharmacy software. Streamline pharmacy operations and improve clinical performance with automated processing, real-time data exchange, and electronic decision support. Download this helpful infographic to: Drive efficiency and patient adherence from referral receipt to delivery and ongoing care – all with our Pharmacy Cloud.

Eye on FDA
NOVEMBER 17, 2020
Coincident to COVID-19 case levels in Europe and the United States entering new and serious levels comes welcome news on the development of a vaccine. Last week Pfizer and BioNTech announced findings from the first interim analysis of the Phase III studies of their candidate vaccine for COVID-19 demonstrating high efficacy levels followed this week by a similar announcement by Moderna.
Pharma Times
NOVEMBER 16, 2020
Janssen will enrol participants in countries with high incidence of COVID-19, including the UK

NY Times
NOVEMBER 20, 2020
A large team of regulators at the agency will take about three weeks to review an application spanning thousands of pages.

pharmaphorum
NOVEMBER 19, 2020
The FDA has agreed to expedite development of two rare disease drugs from Sanofi. The most significant of the two announcements is the Priority Review for avalglucosidase alfa, a potential new therapy for Pompe disease. The review period will be shortened from the standard ten months to six months or less, and the French pharma said the FDA will make the regulatory decision before May 18th.

Outsourcing Pharma
NOVEMBER 18, 2020
A leader from the trial patient services specialist discusses the importance of keeping the participantâs vital role top of mind in research studies.
Pharma Times
NOVEMBER 20, 2020
R&D programme is focused on developing small molecules against SARS-CoV-2 virus
NY Times
NOVEMBER 17, 2020
The Gallup survey held promise for an eventual vaccine rollout, but its authors cautioned that confidence remained lower than it was earlier in the pandemic.

pharmaphorum
NOVEMBER 18, 2020
Pfizer is preparing to file its COVID-19 vaccine after a trial reached the safety milestone required by US regulators for emergency use, according to press reports. The company’s CEO Albert Bourla said that the company is preparing to submit its data to the FDA following last week’s announcement that the vaccine is more than 90% effective, according to an interim analysis of a phase 3 trial.
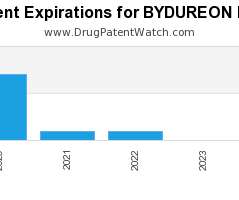
Drug Patent Watch
NOVEMBER 15, 2020
Annual Drug Patent Expirations for BYDUREON+PEN Bydureon Pen is a drug marketed by Astrazeneca Ab and is included in one NDA. It is available from one supplier. There are twenty-nine…. The post New patent for Astrazeneca Ab drug BYDUREON PEN appeared first on DrugPatentWatch - Make Better Decisions.

Pharma Times
NOVEMBER 17, 2020
Companies will develop oral small molecule treatments

NY Times
NOVEMBER 18, 2020
The surge in coronavirus cases worldwide is helping researchers measure more quickly how well their vaccines protect against Covid-19.
pharmaphorum
NOVEMBER 18, 2020
To mark World Antimicrobial Awareness Week NovaBiotics’s Deborah O’Neil tells us why 2021 might be the year anti-infective drugs finally demonstrate their economic value. . COVID-19 has reminded the world how critical infectious disease is, from both a societal and economic standpoint. Cancer is rightly seen as one of the most pressing challenges of our time: but it has not yet caused a multi-trillion hole in the world’s finances.

Outsourcing Pharma
NOVEMBER 18, 2020
A leader from the CRO discusses how the COVID-19 pandemic has accelerated decentralized adoption, and how such trials lead to benefits for participants.

Pharma Times
NOVEMBER 17, 2020
MPs launch inquiry with view of securing extra support for sector

NY Times
NOVEMBER 18, 2020
Lee and Kwame Kwei-Armah will write the movie musical, about the invention of the little blue pill, which will feature music by Stew Stewart and Heidi Rodewald.
pharmaphorum
NOVEMBER 19, 2020
The coronavirus vaccine being developed by Oxford University and AstraZeneca produces a strong immune response in higher risk older patients, a study has found. The phase 2/3 trial results published in The Lancet about the UK-developed shot follow more detailed findings from Pfizer/BioNTech showing that their vaccine protected 94% of adults over 65 years old.

Outsourcing Pharma
NOVEMBER 18, 2020
A leader from the CRO discusses the vital role participants play in a clinical studyâs success, and what it truly means to be patient centric.

Pharma Times
NOVEMBER 18, 2020
Collaboration will focus on developing exosome therapeutics for rare diseases

NY Times
NOVEMBER 16, 2020
Moderna es la segunda compañía que informa de los resultados preliminares de un gran ensayo de una vacuna. Pero todavía faltan meses para que esté ampliamente disponible para el público.

pharmaphorum
NOVEMBER 17, 2020
As regional vice president for Northern Europe & Australia, Simon Lem led the digital launch of Vertex Pharma’s novel cystic fibrosis drug Kaftrio. He tells pharmaphorum about embracing digital change and the need for innovative reimbursement solutions. For Vertex, COVID-19 accelerated a digital transformation much needed for the patients they serve.

Outsourcing Pharma
NOVEMBER 17, 2020
A leader from the global CRO talks about how use of RWE in clinical trials can benefit research sites and sponsors, and satisfy regulatory requirements.
Pharma Times
NOVEMBER 18, 2020
Initial purchase includes 225 million doses of mRNA-based vaccine
NY Times
NOVEMBER 20, 2020
The Nebraska pharmacist was sentenced to nine years and the Maryland drug dealer was sentenced for 14 years for the plot.

pharmaphorum
NOVEMBER 20, 2020
COVID-19 has left us more reliant on media than ever – both the pharma industry and society as a whole. Havas Lynx Group’s 2020 white paper provides a blueprint for pharma brands to inject meaning into their media. “Meaningful media experiences are made by connecting with the right people, in the contexts they spend their time, with content that drives impact.
Drug Patent Watch
NOVEMBER 20, 2020
Annual Drug Patent Expirations for DULERA Dulera is a drug marketed by Merck Sharp Dohme and is included in one NDA. It is available from one supplier. There are two…. The post New patent expiration for Merck Sharp drug DULERA appeared first on DrugPatentWatch - Make Better Decisions.
Let's personalize your content