Lupus discovery lends clues to the fight against long COVID
PharmaVoice
JULY 27, 2022
So far, pharma has yet to provide an answer for long COVID. But award-winning lupus research by Yale professor Akiko Iwasaki help lead the way.

PharmaVoice
JULY 27, 2022
So far, pharma has yet to provide an answer for long COVID. But award-winning lupus research by Yale professor Akiko Iwasaki help lead the way.

pharmaphorum
JULY 26, 2022
Newly diagnosed cancer patients across the UK will get free access to digital mental health therapies, thanks to a new partnership between Macmillan Cancer Support and Big Health. The alliance, billed as the first of its kind in the UK, means that cancer patients will be able to use Big Health apps like Sleepio and Daylight, which aim to treat insomnia and anxiety using cognitive behavioural therapy (CBT) techniques.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Pharma Marketing Network
JULY 27, 2022
In the first two weeks after a cancer diagnosis, there are likely dozens of doctor’s visits, tests, consultations, and decision points. The patient care team supplies an abundance of information, although sifting through brochures and online resources can be daunting for the patient and their caregiver. Adhering to regulatory guidelines is an important step pharma takes to ensure that promotional medical materials contain balanced information that is accurate and relevant. 1 The marketer’s goal

Pharmaceutical Technology
JULY 26, 2022
According to GlobalData’s recent Niemann-Pick Type C (NPC): Opportunity Analysis and Forecast to 2031 report, the NPC market is expected to see significant growth during 2021–31. The three major markets (3MM: the US, Germany and the UK) will increase in market size from $128.35m last year to $188.35m in 2031, at a compound annual growth rate (CAGR) of 3.9%.

Speaker: Chris Antypas and Josh Halladay
Access to limited distribution drugs and payer contracts are key to pharmacy expansion. But how do you prepare your operations to take the next step? Meaningful data: Collect and share clinical data regarding outcomes, utilization, and more Reporting: Limited distribution models require efficient tracking and reporting systems Workflows: Align workflows with specific pharma and payer contractual requirements For in-depth, expert insights on pharmacy expansion, watch this webinar from Inovalon.
PharmaVoice
JULY 28, 2022
The digital medicine company’s chief strategy officer, John Varaklis, explains how its analytic predictive platform could lead to safer and quicker clinical trials.
pharmaphorum
JULY 27, 2022
Pfizer and BioNTech have started a mid-stage trial of a new version of the COVID-19 vaccine based on a version of the spike protein that they hope will offer greater and broader protection against SARS-CoV-2 variants. The vaccine, codenamed BNT162b5, codes for “enhanced” prefusion spike proteins from the original wild-type strain of the virus and the Omicron variant that are designed to increase “the magnitude and breadth of the immune response.” The phase 2 trial will te
Pharmacist Digest brings together the best content for pharmacists from the widest variety of industry thought leaders.

Pharmaceutical Technology
JULY 25, 2022
The COVID-19 pandemic triggered significant changes to the way healthcare operates. Digitalization is now in full swing as medical care integrates with ICT to take the patient experience to the next level. According to Tom Lenaerts, Head of Global Process Engineering at Datwyler, this transformation will reshape the entire healthcare ecosystem. “There are already opportunities for patients to track their health through smart medical devices sending reports directly to their healthcare provider.

PharmaVoice
JULY 25, 2022
A combined venture with Roivant — called Priovant Therapeutics — is looking to be a forerunner in the burgeoning autoimmune disease market.

pharmaphorum
JULY 25, 2022
Sachin Grover, lead of solutions vertical at Philips India, tells me about the company’s innovations during India’s COVID outbreak and how they’ve led to increased healthcare access in the region. There is a vast difference in India’s healthcare system between rural and urban areas, but Philips’ team in the region is working hard to close that gap and improve access to healthcare. “It’s been 75 years since India’s independence.
European Pharmaceutical Review
JULY 25, 2022
The European Commission (EC) has issued a draft proposal for a new EU Regulation on blood, tissues and cells (BTC) to help increase the safety and quality of substances of human origin (SoHO). The Regulation will cover all SoHO (eg, blood, tissues, cells, breast milk and microbiota) except solid organs. A major aim of the proposal is to enhance the protection of recipients of SoHO therapies, as well as donors of SoHO and offspring from medically assisted reproduction.

Advertisement
Are you still using workarounds to manage your daily operations? To achieve peak performance, it's time to explore other options for specialty and infusion pharmacy software. Streamline pharmacy operations and improve clinical performance with automated processing, real-time data exchange, and electronic decision support. Download this helpful infographic to: Drive efficiency and patient adherence from referral receipt to delivery and ongoing care – all with our Pharmacy Cloud.

Outsourcing Pharma
JULY 25, 2022
During the Decentralized Trial Technology event, experts from CVS Health, University of Chicago, and ACRP shared insight on what can increase DCT success.
PharmaVoice
JULY 25, 2022
The latest trial failures in Alzheimer's have shown cracks in the foundation of ingrained theories like amyloid plaque clearance. What will it take to move forward?

Pharmaceutical Technology
JULY 29, 2022
Pfizer has reported a 53% operational growth in revenues to $27.7bn in the second quarter (Q2) of 2022 compared to $18.9bn in the same quarter last year. The rise in revenues was chiefly driven by robust contributions from Covid-19 therapies Paxlovid and Comirnaty. On an operational basis, the revenues rose by $128m or 1%, excluding Paxlovid and Comirnaty’s contributions.

pharmaphorum
JULY 28, 2022
Supply chain shortages have dominated headlines for several months, and the pharma industry has been no exception. While navigating changes has long been a challenge for the industry, the last two years’ events have made supply chain management significantly more difficult. The pandemic effectively shut down supply chain paths overnight, and as a result, pharmaceutical manufacturers have faced delays and bottlenecks, which have caused drug shortages in healthcare systems.

NY Times
JULY 25, 2022
The BA.5 surge is a reminder that we need to take precautions to avoid illness, slow the relentless cycle of new variants and minimize the disruption to our daily lives.

PharmaVoice
JULY 25, 2022
Inozyme Pharma has joined a genomic sequencing program developing a novel rapid test for 388 genetic diseases.
Pharmaceutical Technology
JULY 29, 2022
The Food and Drug Administration (FDA) has acc epted to review ImmunityBio’s Biologics License Application (BLA) for N-803 to treat Bacillus Calmette-Guérin (BCG)-unresponsive non-muscle-invasive bladder cancer (NMIBC) carcinoma in situ (CIS) patients with or without Ta or T1 disease. A decision from the regulatory agency on approval for the treatment is anticipated on 23 May next year.

pharmaphorum
JULY 27, 2022
Health innovation and technology specialist Healthware Group has added another element to its business with the launch of a dedicated medical communications and education division. The new unit – dubbed Healthware MedComms – promises to take a fresh approach to medical communications that meets “the always-on, audience-centric, evidence-driven demands of today’s modern clinical environment.” It adds to Healthware’s fast-growing presence in the healthcare communications se
European Pharmaceutical Review
JULY 29, 2022
Researchers have constructed biohybrid microrobots, by equipping Escherichia coli bacteria with artificial components, that could one day offer a highly targeted and effective cancer treatment option. The team of scientists from the Physical Intelligence Department at the Max Planck Institute for Intelligent Systems combined robotics with E. coli bacteria to construct their innovative drug delivery system.

PharmaVoice
JULY 27, 2022
A trickling launch for Aduhelm and a middling earnings report give Biogen executives a lot to talk about. Is there hope?

Pharmaceutical Technology
JULY 29, 2022
The rate at which children are being identified with autism spectrum disorder (ASD) has tripled in the past two decades. While the prevalence was 1 in 150 children in 2000, it reached the rate of 1 in 44 in 2018, in the US, according to the Centers for Disease Control and Prevention. Some people with ASD can experience acute agitation and crisis behaviours.

pharmaphorum
JULY 27, 2022
Latest UK Life Sciences Competitive Indicators “ought to ring alarm bells across Government,” says ABPI. Despite the UK’s potential as an international life sciences leader, it lags many competitors in a number of key metrics, including access to new medicines and global share of clinical trial recruitment. Richard Torbett, chief executive at the Association of the British Pharmaceutical Industry (ABPI) said Life Sciences Competitive Indicators (LSCI) data published last week “ought to ring alar

Pharma Times
JULY 27, 2022
First carbon minimal pMDI is on track with the joint goal of benefitting patients and the planet

PharmaVoice
JULY 27, 2022
The 2022 president of Women in Bio is equal parts math and gender parity evangelist.
Pharmaceutical Technology
JULY 28, 2022
The Emergency Task Force (ETF) of the European Medicines Agency (EMA) has commenced the review of Veru’s sabizabulin to treat hospitalised Covid-19 patients at increased acute respiratory distress syndrome (ARDS) risk. The review will facilitate the use of the therapy for emergency usage in countries in the European Union (EU). Under this process, the ETF will analyse all available data, including findings from a trial in moderate-to-severe Covid-19 patients admitted to hospital who are at incre
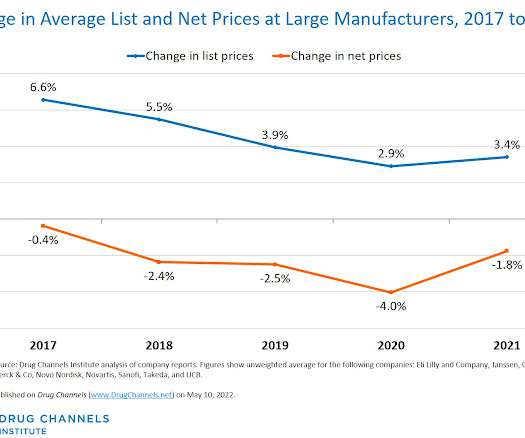
Drug Channels
JULY 28, 2022
This week, I’m rerunning some popular posts while I prepare for this Friday’s live video webinar: Specialty Drugs Update: Trends, Controversies, and Outlook. Click here to see the original post from May 2022. Time for Drug Channels ’ annual update on pricing at the largest pharmaceutical manufacturers. This year’s review includes 10 companies: Eli Lilly and Company, Genentech, GlaxoSmithKline, Janssen, Merck, Novartis, Novo Nordisk, Sanofi, Takeda, and UCB.

Pharma Times
JULY 26, 2022
Funding will develop inhaled therapies using ICOone Nasal in preparation for the next pandemic

European Pharmaceutical Review
JULY 26, 2022
Recommended for approval. Amvuttra * (vutrisiran) was recommended for marketing authorisation as a treatment for adults with hereditary transthyretin-mediated amyloidosis, a rare life-threatening disease that damages multiple nerves across the body. The European Medicines Agency (EMA)’s Committee for Medicinal Products for Human Use (CHMP) adopted a positive opinion for Celdoxome pegylated liposomal (doxorubicin hydrochloride) for the treatment of metastatic breast cancer, advanced ovaria

Pharmaceutical Technology
JULY 28, 2022
Ulcerative colitis (UC), an inflammatory bowel disease, is characterised by inflammation and ulcers in the large intestine, often causing symptoms that impact a patient’s quality of life, including diarrhoea, abdominal pain and cramping, fatigue, weight loss and pain. While the exact cause of UC remains unknown, it is often associated with immune system malfunction.
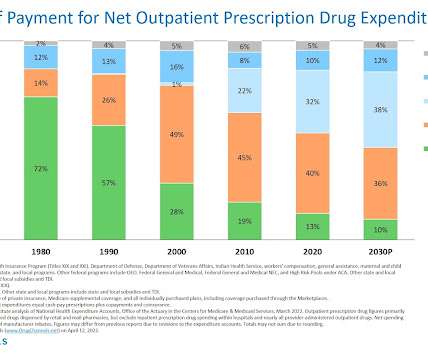
Drug Channels
JULY 29, 2022
This week, I’m rerunning some popular posts while I prepare for today's live video webinar: Specialty Drugs Update: Trends, Controversies, and Outlook. Note that the forecasts below did not account for policy changes that could further reduce pharmaceuticals' share of U.S. healthcare spending. Click here to see the original post from April 2022.

Pharma Times
JULY 26, 2022
Voxelotor is the first medicine in Britain to directly inhibit sickle haemoglobin polymerisation

European Pharmaceutical Review
JULY 29, 2022
Both the European Medicines Agency (EMA) and US Food and Drug Administration (FDA) have endorsed a statement calling for international collaboration on integrating real-world evidence (RWE) into regulatory decision-making. The statement, published by the International Coalition of Medicines Regulatory Authorities (ICMRA), describes how collaboration between regulators and researchers that formed a critical aspect of the world’s COVID-19 response could be leveraged to enhance medicines regulatio

Pharmaceutical Technology
JULY 28, 2022
Bristol Myers Squibb (BMS) has reported a 2% rise in revenues to $11.9bn in the second quarter (Q2) of 2022 compared to $11.7bn in Q2 last year. The revenue growth was driven by in-line products, mainly Eliquis and Opdivo, as well as the company’s latest product portfolio comprising Abecma, Opdualag and Reblozyl. New LOE products, especially Revlimid and foreign exchange, impacts partially offset the increase in revenues.
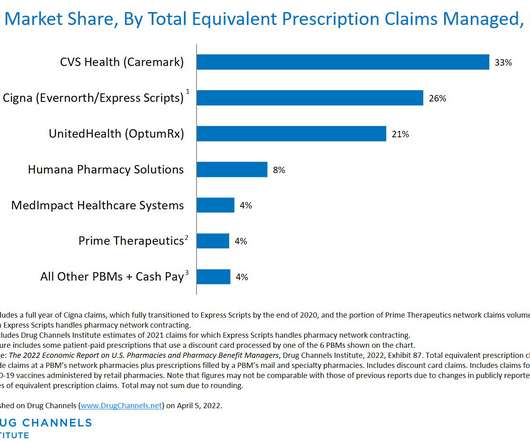
Drug Channels
JULY 25, 2022
This week, I’m rerunning some popular posts while I prepare for this Friday’s live video webinar: Specialty Drugs Update: Trends, Controversies, and Outlook. Click here to see the original post from April 2022. The highly-consolidated PBM market notched another year of increased concentration. Consider Drug Channels Institute's latest estimates of pharmacy benefit manager (PBM) market share, shown in the chart below.

Pharma Times
JULY 27, 2022
Predicting which women are likely to develop pre-eclampsia will help clinicians to decide on the most appropriate care
Let's personalize your content