Grünenthal’s non-opioid pain drug gets breakthrough tag
pharmaphorum
MAY 23, 2023
Grünenthal’s non-opioid pain drug gets breakthrough tag Phil.Taylor Tue, 05/23/2023 - 08:20 Bookmark this

 tag pain
tag pain 
pharmaphorum
MAY 23, 2023
Grünenthal’s non-opioid pain drug gets breakthrough tag Phil.Taylor Tue, 05/23/2023 - 08:20 Bookmark this
STAT
DECEMBER 12, 2023
These drugs have price tags of $2.2 The news comes as researchers are still processing the Food and Drug Administration’s landmark approval of two cutting-edge sickle cell therapies, one made by Vertex Pharmaceuticals and CRISPR Therapeutics and the other by Bluebird Bio. million and $3.1 million, respectively.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

PharmaShots
MAY 19, 2023
Alimera Acquires Rights from EyePoint Pharmaceuticals to Commercialise Yutiq in the US Date: May 19, 2023 | Tags: Alimera, EyePoint Pharmaceuticals, Yutiq, Iluvien, chronic non-infectious uveitis, US, Pharma Sobi Reports EMA’s Validation of MAA for Efanesoctocog Alfa to Treat Haemophilia A Date: May 19, 2023 | Tags: Sobi, Efanesoctocog Alfa, (..)

PharmaShots
MAY 12, 2023
Sanofi Reports P-IIIb Trial (HARMONIE) Results of Nirsevimab for the Prevention of Hospitalizations due to RSV-Related LRTD Date: May 12, 2023 | Tags: Sanofi, Nirsevimab, RSV-Related LRTD, Clinical Trial, P-IIIb, HARMONIE Trial G1 Therapeutics Presents Preliminary Results from P-II Trial of Trilaciclib for Triple-Negative Breast Cancer at ESMO 2023 (..)

PharmaShots
MARCH 17, 2023
5-Adapted Bivalent Booster to Treat COVID-19 in Children ≤5 Years Date: Mar 15, 2023 | Tags: Pfizer, BioNTech, Omicron BA.4/BA.5-Adapted 5-Adapted Bivalent Booster to Treat COVID-19 in Children ≤5 Years Date: Mar 15, 2023 | Tags: Pfizer, BioNTech, Omicron BA.4/BA.5-Adapted

PharmaShots
FEBRUARY 3, 2023
4D Molecular Therapeutics Receives the US FDA’s IND Clearance of 4D-150 for the Treatment of Diabetic Macular Edema Date: Feb 03, 2023 | Tags: 4D Molecular Therapeutics, 4D-150, Diabetic Macular Edema, Regulatory, US, FDA, IND AstraZeneca and Amgen Receive the US FDA’s Approval of Tezspire (tezepelumab) for the Treatment of Severe Asthma (..)
Pharmaceutical Technology
DECEMBER 5, 2022
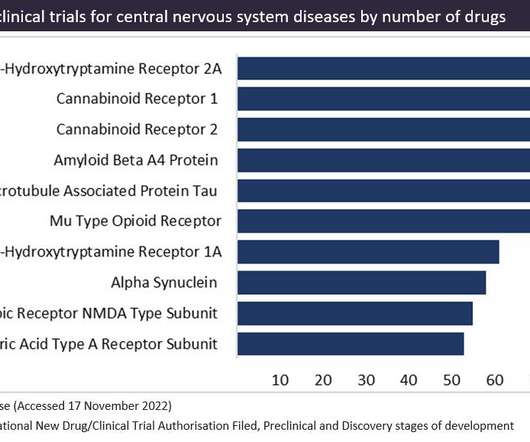
Cannabinoid receptors are a popular therapeutic target for cannabinoid-based drugs in the treatment of pain, neurological disorders and inflammation, according to GlobalData’s Pharma Intelligence Centre Drugs database. This is followed by epilepsy at 8% of the pipeline and multiple sclerosis at 7% of the pipeline.
Let's personalize your content