Behind the new topical treatment set to disrupt the psoriasis market
PharmaVoice
JULY 11, 2022
Phil Brown, Dermavant’s chief medical officer, discusses VTAMA — the first topical cream approved for psoriasis in 25 years.

 topic
topic 
PharmaVoice
JULY 11, 2022
Phil Brown, Dermavant’s chief medical officer, discusses VTAMA — the first topical cream approved for psoriasis in 25 years.
RX Note
JULY 15, 2023
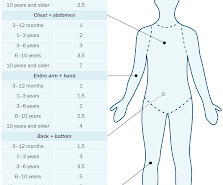
Introduction Topical corticosteroids have a major role in the management of many skin diseases. The potency classification of topical corticosteroid above is extracted from Australian Medicines Handbook , which potency estimate is based on vasoconstrictive ability, inhibition and potential for causing adverse effects.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

The Pharmacist
JANUARY 9, 2024
If elected, these MPs would campaign […] The post Make pharmacy an election topic, urges RPS appeared first on The Pharmacist. It has developed a pledge, dubbed a ‘Pharmacy Manifesto’, asking MPs to sign a ‘prescription for the nation’s health’.
Welltopia Pharmacy
JANUARY 17, 2024
Topical Metformin: A New Era in Diabetes Care Understanding Diabetes: Types and Prevalence Diabetes, a chronic condition affecting millions worldwide,

Express Pharma
AUGUST 1, 2023
Fluocinolone Acetonide Topical Oil (RLD Derma-Smoothe/FS) had estimated annual sales of $9 million in the US. Fluocinolone Acetonide Topical Oil (RLD Derma-Smoothe/FS) had estimated annual sales of $9 million in the US. The post Lupin US FDA approval for Fluocinolone Acetonide Topical Oil appeared first on Express Pharma.
RX Note
FEBRUARY 26, 2023
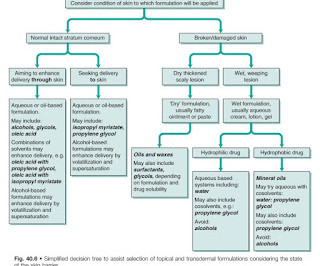
It has been estimated that for topical products such as creams and gels, typically only between 1% and 3% of the applied dose is bioavailable. NOTE: Topical treatments used for local effects have a decreased incidence of systemic side effects and generally provide faster relief. Factors affecting formulation type of topical therapy.

Pharmaceutical Technology
MAY 26, 2023
Krystal Biotech has received approval from the US Food and Drug Administration for topical gene therapy VYJUVEK to treat dystrophic epidermolysis bullosa (DEB) in adults and in children aged six months and above. Topic sponsors are not involved in the creation of editorial content.
Let's personalize your content