Clinical trial shows rheumatoid arthritis drug could prevent disease
World Pharma News
FEBRUARY 14, 2024
A drug used to treat rheumatoid arthritis could also prevent the disease in individuals deemed to be at risk.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 clinical rheumatoid-arthritis
clinical rheumatoid-arthritis 
World Pharma News
FEBRUARY 14, 2024
A drug used to treat rheumatoid arthritis could also prevent the disease in individuals deemed to be at risk.

STAT
DECEMBER 11, 2023
During 2022, drugmakers substantially raised prices on eight widely used medicines without any new clinical evidence to justify the increases, leading patients and health insurers in the U.S. to spend an additional $1.2 billion last year, according to a new report.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

STAT
OCTOBER 4, 2023
The Center charges that these tests are not nearly as accurate as their maker claims they are, and that they aren’t good at distinguishing between fibromyalgia and other diseases with similar symptoms such as rheumatoid arthritis and lupus. The suit cites a STAT investigation of EpicGenetics published in October 2021.

Big Molecule Watch
APRIL 26, 2024
On April 23, 2024, Alvotech announced “positive topline results from a confirmatory clinical study for AVT05,” Alvotech’s proposed golimumab biosimilar to Janssen Biotech, Inc.’s Following the confirmatory clinical results, Alvotech announced its intention to file marketing applications for AVT05 later this year.
European Pharmaceutical Review
APRIL 30, 2024
CHMP biosimilar recommendations A positive opinion for Tofidence (tocilizumab) was adopted by the committee, indicated for rheumatoid arthritis, COVID-19, polyarticular juvenile idiopathic arthritis, and systemic juvenile idiopathic arthritis.

RX Note
MARCH 22, 2024
Introduction Rheumatoid arthritis is a chronic autoimmune disease characterized by persistent synovitis, systemic inflammation and the presence of autoantibodies. Clinical remission should be the primary treatment target , but low disease activity may be appropriate for patients with long-standing disease or comorbidities.
ALiEM - Pharm Pearls
OCTOBER 20, 2023
It may present as the first clinical finding of or a precursor to diabetes, although its course is unaffected by glycemic control and it is unrelated to other diabetic complications including renal, ocular, and vascular problems. It has been associated with thyroid disease, inflammatory bowel disease, rheumatoid arthritis, and sarcoidosis.

Big Molecule Watch
MAY 19, 2023
The clinical study will compare the efficacy, safety, and immunogenicity of AVT05 and Simponi in adult patients with moderate to severe rheumatoid arthritis. On May 15, 2023, Boan Biotech announced the completion of patient enrollment for a Phase III clinical trial of the company’s Dulaglutide Injection (BA5101) in China.

Pharmaceutical Technology
OCTOBER 26, 2023
Tocilizumab biosimilar is under clinical development by Fresenius Kabi SwissBioSim and currently in Pre-Registration for Rheumatoid Arthritis.

Pharmaceutical Technology
DECEMBER 22, 2023
Imvotamab is under clinical development by IGM Biosciences and currently in Phase I for Rheumatoid Arthritis.

Pharmaceutical Technology
FEBRUARY 22, 2023
Tocilizumab biosimilar is under clinical development by Bio-Thera Solutions and currently in Pre-Registration for Rheumatoid Arthritis.
European Pharmaceutical Review
JANUARY 4, 2023
EVOQ Therapeutics Inc has agreed to license its NanoDisc technology for rheumatoid arthritis (RA) and lupus to Gilead Sciences, Inc, enabling EVOQ to potentially receive up $658.5 The companies will collaborate to advance preclinical development and Gilead will be responsible for clinical development and commercialisation.

Med Ed 101
JULY 31, 2022
The most common use in clinical practice that I’ve seen sulfasalazine used for is rheumatoid arthritis, but it can also play a role in ulcerative colitis and Crohn’s disease. In this post, I’ll discuss important sulfasalazine clinical pearls and […].
pharmaphorum
OCTOBER 12, 2021
Usually used to treat rheumatoid arthritis, Actemra was granted emergency approval in June for hospitalised patients 2 years of age and older who are receiving systemic corticosteroids and need respiratory support after it was shown to reduce mortality in a clinical trial.

European Pharmaceutical Review
APRIL 3, 2023
It relies partly on results of pre-clinical tests and clinical trials of an already authorised reference product and in part on new data. This condition is a persistent swelling which does not respond to diuretics and sodium restriction. This medicine was submitted as a hybrid application.

Pharmaceutical Technology
MARCH 2, 2023
Rituximab biosimilar is under clinical development by Dr. Reddy’s Laboratories and currently in Phase III for Rheumatoid Arthritis. According to GlobalData, Phase III drugs for Rheumatoid Arthritis have a 65% phase transition success rate (PTSR) indication benchmark for progressing into Pre-Registration.

pharmaphorum
DECEMBER 15, 2021
Bristol-Myers Squibb’s rheumatoid arthritis drug Orencia has been approved by the FDA to prevent graft versus host disease (GvHD), a serious complication of haematopoietic stem cell transplant (HSCT) used to treat leukaemias and other blood cancers. billion in the first nine months of the year.

European Pharmaceutical Review
NOVEMBER 1, 2022
The proposed restrictions are due to a review of available data, including clinical trial results for Xeljanz and initial findings from an observational study of Olumiant. The PRAC has confirmed that individuals with the following chronic inflammatory disorders should not take JAK inhibitors: Rheumatoid arthritis (RA).
European Pharmaceutical Review
JANUARY 4, 2023
EVOQ Therapeutics Inc has agreed to license its NanoDisc technology for rheumatoid arthritis (RA) and lupus to Gilead Sciences, Inc, enabling EVOQ to potentially receive up $658.5 The companies will collaborate to advance preclinical development and Gilead will be responsible for clinical development and commercialisation.
European Pharmaceutical Review
DECEMBER 7, 2023
A world-first clinical trial in type 1 diabetes In the Phase II clinical trial for baricitinib, investigators monitored the blood glucose and insulin production of 91 participants aged between 10 and 30 years old, over a one-year period.
Pharmaceutical Technology
MAY 25, 2023
Yuflyma is indicated to treat juvenile idiopathic arthritis, hidradenitis suppurativa, Crohn’s disease, plaque psoriasis, ankylosing spondylitis, rheumatoid arthritis, psoriatic arthritis and ulcerative colitis.

Pharmacy Times
JUNE 2, 2023
Upadacitinib 15 mg once daily had a better clinical response compared to adalimumab 40 mg every other week at 12 weeks in 3-year follow-up data among patients with rheumatoid arthritis.
European Pharmaceutical Review
MAY 18, 2023
The NICE recommendation is based on data from three Phase III clinical trials, which demonstrated significantly more patients treated with upadacitinib achieved the co-primary endpoints of clinical remission and endoscopic response.
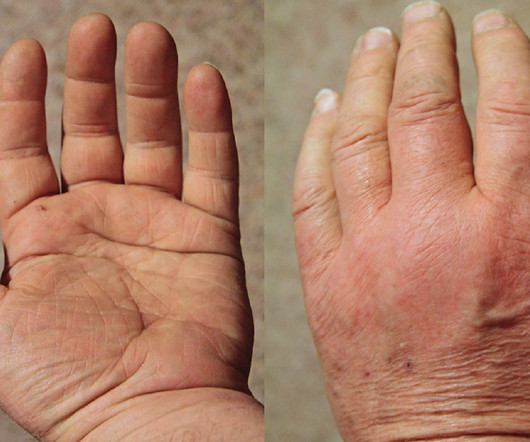
Pharmaceutical Technology
MARCH 1, 2023
It has also been approved in several countries for treating moderate-to-severe active rheumatoid arthritis (RA) in adult patients who have not responded to or tolerated previous therapy. Developed using VelocImmune technology of Regeneron, Kevzara binds to the IL-6 receptor specifically and inhibits IL-6-mediated signalling.

European Pharmaceutical Review
NOVEMBER 22, 2022
a China-based pharma company has announced it has received clearance for its Investigational New Drug (IND) application from the US Food and Drug Administration (FDA) to initiate clinical studies of T21, a 3D-printed medicine that can target specific segments in the colon to more safely deliver oral ulcerative colitis (UC) drugs.

Pharmaceutical Technology
FEBRUARY 19, 2023
Annexuzlimab is under clinical development by MedAnnex and currently in Phase I for Kidney Cancer (Renal Cell Cancer). Attributes of the drug, company and its clinical trials play a fundamental role in drug-specific PTSR and likelihood of approval. It also provides non-clinical testing programs.

PharmaShots
APRIL 28, 2023
Shots: Eli Lilly has licensed ZB-106 (Tibulizumab) to Zura, and in exchange, Zura will pay Eli Lilly $4.25 million in Class A Ordinary Shares as part of the upfront cash payment Zura has also reported its agreement to sell ~18.8M Class A ordinary shares at $4.25

PharmaShots
APRIL 28, 2023
In Aug 2022, the company received IND clearance from the USFDA to initiate the P-I clinical trial of DWP213388. Also, Vitalli Bio will be granted an option to license earlier-stage dual-target inhibitors in development by Daewoong DWP213388, a BTK and ITK inhibitor.

PharmaShots
MAY 4, 2023
rheumatoid arthritis The primary outcome measures will be to show the comparative efficacy of AVT05 vs Simponi @16wks. based on change from baseline on a standardized DAS28-CRP which evaluates the severity of RA using clinical and laboratory data Additionally, the company initiates PK study for AVT05 in Jan 2023.

Pharmaceutical Technology
NOVEMBER 1, 2022
These drugs could potentially treat GBA and some inflammatory ailments, including irritable bowel syndrome, inflammatory bowel diseases, rheumatoid arthritis, atopic dermatitis, oligoarticular juvenile idiopathic arthritis and autism spectrum disorder.

PharmaShots
JUNE 27, 2023
Food and Drug Administration (FDA) have accepted the Marketing Authorization Application and the abbreviated Biologics License Application (aBLA), respectively, for BIIB800 in Q4 2022 with the Phase 3 trial being conducted in rheumatoid arthritis (RA) patients. Smriti: Can we talk about BIIB800, a tocilizumab biosimilar?

pharmaphorum
DECEMBER 6, 2021
Therefore extensive consideration of the clinical and cost-effectiveness of treatments must be made, to justify their value for money and their value to the health system. The combination treatment issue. The post How the combination treatment challenge impacts patients and how we are working together to solve it appeared first on.

pharmaphorum
JANUARY 15, 2021
Join the world’s top ILD specialists as they guide you through the seminal research and insights you need to confidently define, understand, and develop clinically effective antifibrotic therapies against connective tissue disease, rheumatoid arthritis, hypersensitivity pneumonitis, Goodpasture’s syndrome, and much more.

European Pharmaceutical Review
MARCH 4, 2024
This growth is accompanied by a substantial number of clinical trials, with close to 20,000 active investigations, involving about 9,500 new biologic treatments. Despite issues of potential toxicity and biocompatibility, several polymers have advanced to clinical trial stages. percent through 2027.

Pharmaceutical Technology
MARCH 21, 2023
Gilead Sciences has exercised its option to exclusively license the investigational targeted protein degrader development candidate, NX‑0479, from clinical-stage biopharmaceutical company Nurix Therapeutics. The company stated that the degradation of IRAK4 can help treat rheumatoid arthritis (RA), as well as other inflammatory diseases.

European Pharmaceutical Review
SEPTEMBER 20, 2022
Over the past 15 years and the experience from clinical practice has shown that in terms of efficacy, safety and immunogenicity they are comparable to their reference products and are therefore interchangeable,” commented EMA’s Executive Director, Emer Cooke.

pharmaphorum
SEPTEMBER 2, 2021
The FDA also said the risks seemed to be higher with both higher and lower doses of Xeljanz/Xeljanz XR, whereas an earlier assessment suggested only implicated the higher dose of the drugs, used to treat rheumatoid arthritis and ulcerative colitis. The result?

Pharmaceutical Technology
FEBRUARY 25, 2023
ASP-1570 is under clinical development by Astellas Pharma and currently in Phase II for Metastatic Melanoma. Attributes of the drug, company and its clinical trials play a fundamental role in drug-specific PTSR and likelihood of approval.

Pharmaceutical Technology
FEBRUARY 25, 2023
ASP-1570 is under clinical development by Astellas Pharma and currently in Phase II for Solid Tumor. Attributes of the drug, company and its clinical trials play a fundamental role in drug-specific PTSR and likelihood of approval.

Pharmaceutical Technology
JULY 27, 2022
Clinical remission according to Adapted Mayo Score was the primary endpoint of the studies while secondary endpoints comprised clinical response and mucosal healing among others. According to the findings, the trials met the primary endpoint and all secondary endpoints with statistical significance.

pharmaphorum
JULY 1, 2021
The German drugmaker will work with startup neuroloop on bioelectronics , put simply the harnessing of electrical stimulation to treat human disease, with a focus on chronic conditions like arthritis and inflammatory bowel disease.

Pharmaceutical Technology
FEBRUARY 27, 2023
Hydrocortisone is under clinical development by Antares Pharma and currently in Phase I for Adrenal Insufficiency. Attributes of the drug, company and its clinical trials play a fundamental role in drug-specific PTSR and likelihood of approval. It also develops products to reduce the risk of preterm birth in pregnant women.
Pharmaceutical Technology
NOVEMBER 10, 2022
The regulatory agency granted EUA based on findings from the double-blind, randomised, controlled Phase III SAVE-MORE clinical trial of Kineret. In the country, Kineret is intended for lowering signs and symptoms and slowing structural damage progression in patients with moderately to severely active rheumatoid arthritis (RA).

pharmaphorum
OCTOBER 27, 2022
GSK has said it will not be able to move ahead with regulatory filings for otilimab as a treatment for rheumatoid arthritis (RA), after it failed to show the desired level of efficacy in two phase 3 trials. The post GSK shelves arthritis blockbuster hope otilimab after lacklustre data appeared first on.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content